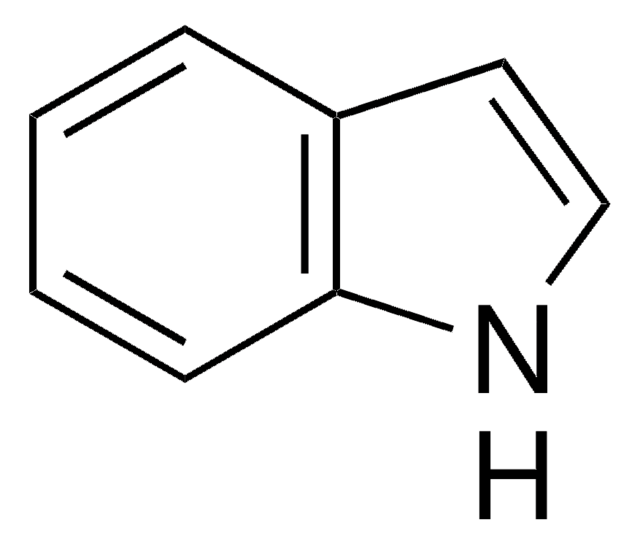
推薦產品
品質等級
化驗
98%
mp
202-206 °C (lit.)
SMILES 字串
OC(=O)c1cc2ccccc2[nH]1
InChI
1S/C9H7NO2/c11-9(12)8-5-6-3-1-2-4-7(6)10-8/h1-5,10H,(H,11,12)
InChI 密鑰
HCUARRIEZVDMPT-UHFFFAOYSA-N
基因資訊
human ... SRD5A1(6715)
rat ... Grin2a(24409)
尋找類似的產品? 前往 產品比較指南
應用
- (±)-二溴谷胱甘肽和类似物全合成的反应物
- 吡咯里西啶生物碱(±)-颈花脒合成的反应物
- 肾素霉素G类似物立体选择性制备的反应物
- 通过还原吲哚-2-羧酸以及随后的氧化、缩合、还原、酰胺化和Kharasch自由基环化进行螺氧吲哚吡咯烷制备的反应物
- Pd催化的环化反应反应物
- N,N′-(戊烷)二基双[吲哚甲酰胺]和N,N′-[亚苯基双(亚甲基)]双[吲哚甲酰胺]衍生物制备的反应物
儲存類別代碼
11 - Combustible Solids
水污染物質分類(WGK)
WGK 3
閃點(°F)
Not applicable
閃點(°C)
Not applicable
個人防護裝備
Eyeshields, Gloves, type N95 (US)
客戶也查看了
T Tonohiro et al.
General pharmacology, 28(4), 555-560 (1997-04-01)
1. A putative agonist for the strychnine-sensitive glycine receptor picolinic acid was tested for its anticonvulsant activities in mice and muscle-relaxant activities in rats and compared with indole-2-carboxylic acid (I2CA), an antagonist for the strychnine-insensitive glycine receptor. Their effects on
James F Dropinski et al.
Bioorganic & medicinal chemistry letters, 15(22), 5035-5038 (2005-09-13)
A series of novel aryl indole-2-carboxylic acids has been identified as potent selective PPARgamma modulators. Their chemical synthesis and in vitro activities are discussed. Compound 5 was selected for in vivo testing in the db/db mouse model of type 2
R Rama Suresh et al.
The Journal of organic chemistry, 77(16), 6959-6969 (2012-07-26)
Two methodologies, one involving Ar-I reactivity and the other through C-H functionalization, for the formation of indolo[2,3-c]pyrane-1-ones via the corresponding allenes, are presented. A highly efficient approach to indolo[2,3-c]pyrane-1-one derivatives through the Pd-catalyzed regioselective annulation of allenes with 3-iodo-1-alkylindole-2-carboxylic acids
Hideyuki Shiozawa et al.
Journal of the American Chemical Society, 124(15), 3914-3919 (2002-04-11)
Glycopeptide antibiotics of the vancomycin group bind to bacterial cell wall analogue precursors, and typically also form dimers. We have studied the interplay between these two sets of noncovalent bonds formed at separate interfaces. Indole-2-carboxylic acid (L) forms a set
Gopinadhan N Anilkumar et al.
Bioorganic & medicinal chemistry letters, 21(18), 5336-5341 (2011-08-16)
SAR development of indole-based palm site inhibitors of HCV NS5B polymerase exemplified by initial indole lead 1 (NS5B IC(50)=0.9 μM, replicon EC(50)>100 μM) is described. Structure-based drug design led to the incorporation of novel heterocyclic moieties at the indole C3-position
我們的科學家團隊在所有研究領域都有豐富的經驗,包括生命科學、材料科學、化學合成、色譜、分析等.
聯絡技術服務