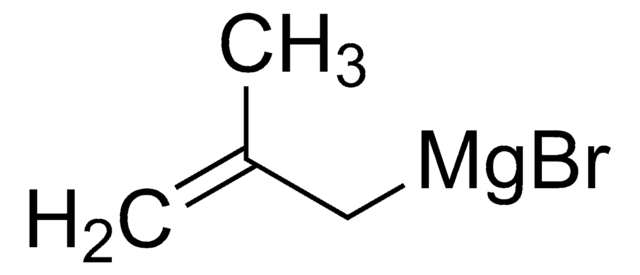
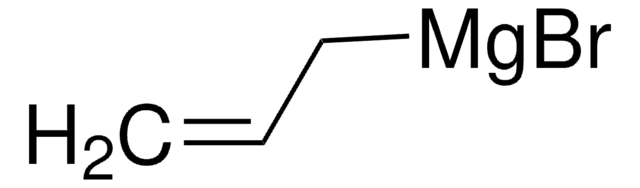
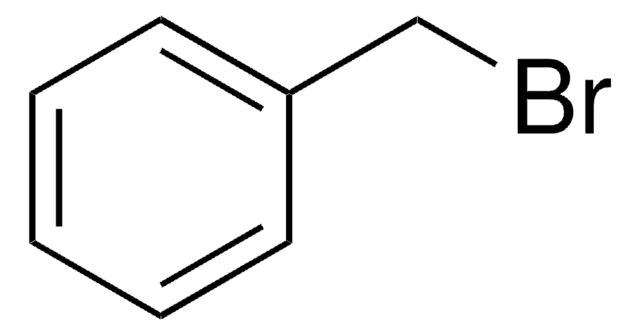
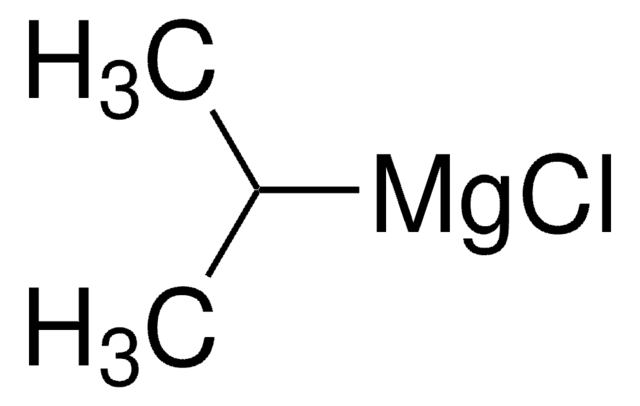
推薦產品
品質等級
化驗
95%
mp
46-51 °C (lit.)
官能基
bromo
iodo
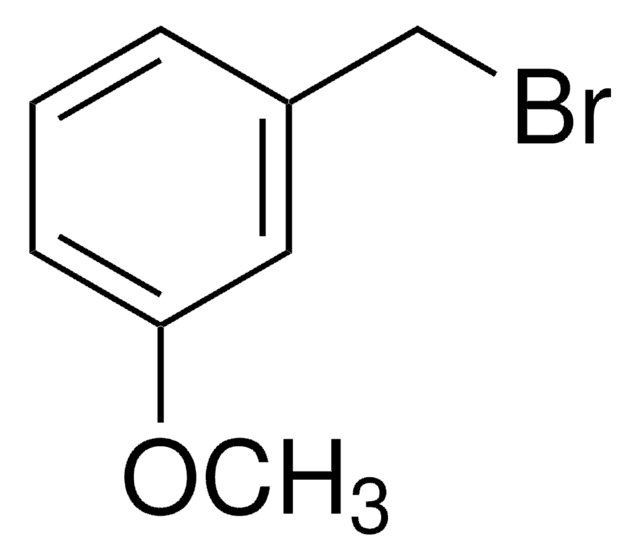
SMILES 字串
BrCc1cccc(I)c1
InChI
1S/C7H6BrI/c8-5-6-2-1-3-7(9)4-6/h1-4H,5H2
InChI 密鑰
BACZSVQZBSCWIG-UHFFFAOYSA-N
一般說明
3-Iodobenzyl bromide (3-IBBr, m-iodobenzyl bromide) is a meta-isomer of iodobenzyl bromide. It can be synthesized by the bromination of m-iodotoluene.
應用
3-Iodobenzyl bromide (3-IBBr, m-iodobenzyl bromide) was used as a derivatization reagent for the extraction and purification of thiouracil (TU).
It may be used in the synthesis of the following:
It may be used in the synthesis of the following:
- meta-substituted phenylalanine derivatives
- N6-substituted aristeromycin derivative
- (N)-methanocarba-N6-(3-iodobenzyl)adenosine
訊號詞
Danger
危險聲明
危險分類
Eye Dam. 1 - Skin Corr. 1B
儲存類別代碼
8A - Combustible corrosive hazardous materials
水污染物質分類(WGK)
WGK 3
閃點(°F)
235.4 °F - closed cup
閃點(°C)
113 °C - closed cup
個人防護裝備
Eyeshields, Faceshields, Gloves, type P3 (EN 143) respirator cartridges
Julie A L Kiebooms et al.
Applied and environmental microbiology, 80(23), 7433-7442 (2014-09-28)
In recent years, the frequent detection of the banned thyreostat thiouracil (TU) in livestock urine has been related to endogenous TU formation following digestion of glucosinolate-rich Brassicaceae crops. Recently, it was demonstrated that, upon in vitro digestion of Brassicaceae, fecal
Barbara Woźniak et al.
Journal of veterinary research, 62(4), 511-517 (2019-02-08)
In the European Union, the use of thyreostatic drugs for fattening slaughter animals has been banned since 1981 under Council Directive 81/602/EEC. For protection of consumer health against unwanted residues and in compliance with Directive 96/23, each EU country must
Reaction of some sulfonyl halides with trypsin.
Kuranova IP and Konareva NV.
Bulletin of the Academy of Sciences of the USSR, Division of chemical science, 22(10), 2251-2254 (1973)
K A Jacobson et al.
Journal of medicinal chemistry, 43(11), 2196-2203 (2000-06-08)
Adenosine receptor agonists have cardioprotective, cerebroprotective, and antiinflammatory properties. We report that a carbocyclic modification of the ribose moiety incorporating ring constraints is a general approach for the design of A(1) and A(3) receptor agonists having favorable pharmacodynamic properties. While
Phase Transfer Catalyst (PTC) Catalyzed Alkylations of Glycinamides for Asymmetric Syntheses of alpha-Amino Acid Derivatives.
Park YS, et al.
Bull. Korean Chem. Soc., 22(9), 958-962 (2001)
我們的科學家團隊在所有研究領域都有豐富的經驗,包括生命科學、材料科學、化學合成、色譜、分析等.
聯絡技術服務