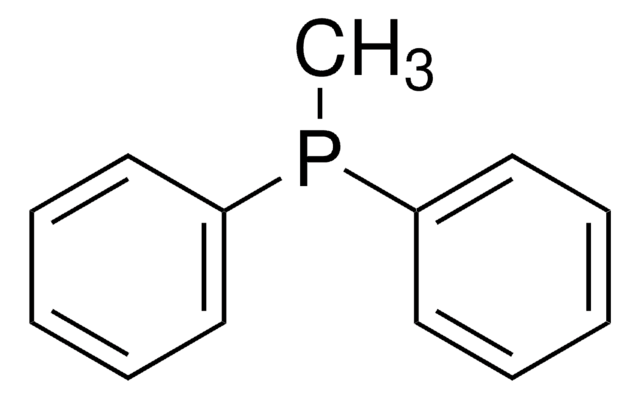
推薦產品
品質等級
化驗
97%
反應適用性
core: iridium
reagent type: catalyst
mp
224 °C (dec.) (lit.)
SMILES 字串
[Ir+].F[P-](F)(F)(F)(F)F.C1CC=CCCC=C1.CP(c2ccccc2)c3ccccc3.CP(c4ccccc4)c5ccccc5
InChI
1S/2C13H13P.C8H12.F6P.Ir/c2*1-14(12-8-4-2-5-9-12)13-10-6-3-7-11-13;1-2-4-6-8-7-5-3-1;1-7(2,3,4,5)6;/h2*2-11H,1H3;1-2,7-8H,3-6H2;;/q;;;-1;+1/b;;2-1-,8-7-;;
InChI 密鑰
LXKHQEQLBSJJCO-JXNOXZOESA-N
相關類別
應用
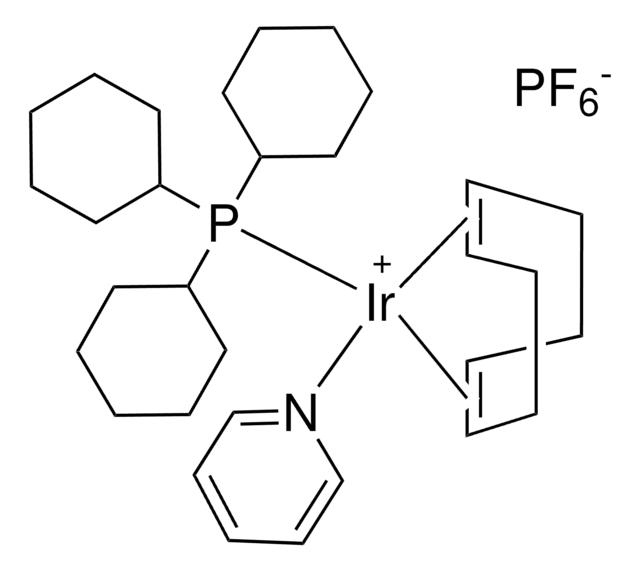
(1,5-Cyclooctadiene)bis(methyldiphenylphosphine)iridium(I) hexafluorophosphate is an air-stable cationic iridium complex, generally employed in allyl isomerization reactions. Upon bubbling hydrogen into the solution of [Ir(cod)(PPh2Me)2]PF6, preferably in tetrahydrofuran, results in the formation of active catalyst [IrH2(solv)2(PPh2Me)2]PF6 which can be used in the isomerization of 3-(silyloxy)-1-propenylboronates to (γ-(silyloxy)allyl)boronic esters; diallyl ethers to allyl (E)-vinyl ethers and 1-alkenylboronates to corresponding 3-alkoxyallylboronates.
訊號詞
Warning
危險分類
Acute Tox. 4 Dermal - Acute Tox. 4 Inhalation - Acute Tox. 4 Oral - Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
標靶器官
Respiratory system
儲存類別代碼
11 - Combustible Solids
水污染物質分類(WGK)
WGK 3
閃點(°F)
Not applicable
閃點(°C)
Not applicable
個人防護裝備
dust mask type N95 (US), Eyeshields, Gloves
客戶也查看了
Paulsen, H.; Adermann, K.
Carbohydrate Research, 175, 163-163 (1988)
Synthesis of Chiral Esters of (E)-3-(Silyloxy)-2-propenylboronic Acid via the Iridium-Catalyzed Isomerization of the Double Bond.
Yamamoto Y, et al.
The Journal of Organic Chemistry, 64(1), 296-298 (1999)
Stereoselective Isomerization of Unsymmetrical Diallyl Ethers to Allyl (E)-Vinyl Ethers by a Cationic Iridium Catalyst.
Yamamoto Y, et al.
Synthetic Communications, 30(13), 2383-2391 (2000)
Intramolecular allylboration of γ-(?-formylalkoxy) allylboronates for syntheses of trans-or cis-2-(ethenyl) tetrahydropyran-3-ol and 2-(ethenyl) oxepan-3-ol.
Yamamoto Y, et al.
Tetrahedron, 59(4), 537-542 (2003)
Oltvoort, J. J.; Van Boeckel, C. A. A. et al.
Synthesis, 305-305 (1981)
Global Trade Item Number
| 庫存單位 | GTIN |
|---|---|
| 337455-500MG | 4061826743584 |
| 337455-50MG | 4061833279502 |
我們的科學家團隊在所有研究領域都有豐富的經驗,包括生命科學、材料科學、化學合成、色譜、分析等.
聯絡技術服務


![[1,1′-双(二苯基膦)二茂铁]二氯化钯(II)](/deepweb/assets/sigmaaldrich/product/structures/130/734/8846aa26-1858-458a-998d-8c306c13bf0f/640/8846aa26-1858-458a-998d-8c306c13bf0f.png)