推荐产品
一般說明
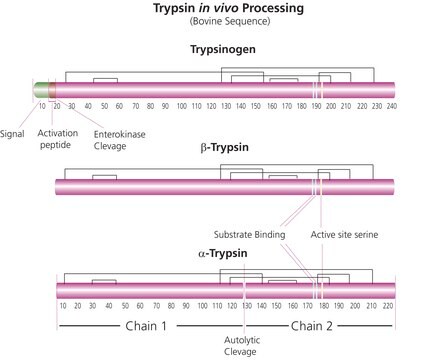
Trypsin is routinely used in proteomics research for peptide mapping and protein sequence work due to its highly specific cleavage resulting in a limited number of tryptic peptides. Trypsin is a pancreatic serine endoprotease which hydrolyzes peptide bonds specifically at the carboxyl side of arginine and lysine residues. The rate of hydrolysis is slower if an acidic residue is on either side of the cleavage site and cleavage may not occur if a proline residue is on the carboxyl side. The enzyme also exhibits esterase and amidase activities. Trypsin has an average molecular mass of 23.29 kDa and a pH optimum near 8.0.
This product is prepared from recombinant trypsin, porcine sequence and the lysine residues have been dimethylated to further restrict autolysis. It is naturally devoid of chymotryptic activity. This high quality trypsin is suitable for proteomics use.
Specific activity: >= 10,000 BAEE units per mg protein.
This product is prepared from recombinant trypsin, porcine sequence and the lysine residues have been dimethylated to further restrict autolysis. It is naturally devoid of chymotryptic activity. This high quality trypsin is suitable for proteomics use.
Specific activity: >= 10,000 BAEE units per mg protein.
訊號詞
Danger
危險分類
Aquatic Chronic 2 - Eye Dam. 1 - Met. Corr. 1 - Resp. Sens. 1 - Skin Corr. 1A - Skin Sens. 1 - STOT SE 3
標靶器官
Respiratory system
安全危害
儲存類別代碼
8A - Combustible corrosive hazardous materials
水污染物質分類(WGK)
WGK 3
閃點(°F)
Not applicable
閃點(°C)
Not applicable
其他客户在看
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系技术服务部门