推荐产品
等級
certified reference material
pharmaceutical secondary standard
品質等級
agency
traceable to BP 553
traceable to Ph. Eur. P2900000
traceable to USP 1559006
API 家族
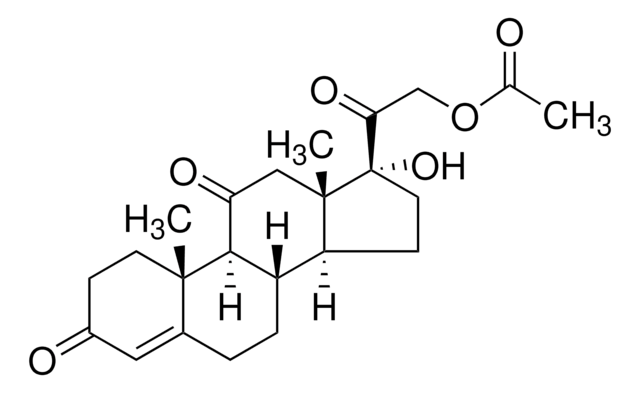
prednisone
CofA
current certificate can be downloaded
技術
HPLC: suitable
gas chromatography (GC): suitable
mp
236-238 °C (lit.)
應用
pharmaceutical (small molecule)
形式
neat
儲存溫度
2-30°C
SMILES 字串
O=C1C=C[C@@]2(C)C(CC[C@]([C@@](CC[C@@]3(C(CO)=O)O)([H])[C@]3(C)C4)([H])[C@]2([H])C4=O)=C1
InChI
1S/C21H26O5/c1-19-7-5-13(23)9-12(19)3-4-14-15-6-8-21(26,17(25)11-22)20(15,2)10-16(24)18(14)19/h5,7,9,14-15,18,22,26H,3-4,6,8,10-11H2,1-2H3/t14-,15-,18+,19-,20-,21-/m0/s1
InChI 密鑰
XOFYZVNMUHMLCC-ZPOLXVRWSA-N
基因資訊
human ... NR3C1(2908)
正在寻找类似产品? 访问 产品对比指南
一般說明
Pharmaceutical secondary standards for application in quality control, provide pharma laboratories and manufacturers with a convenient and cost-effective alternative to the preparation of in-house working standards.
應用
Prednisone may be used as a pharmaceutical reference standard for the determination of the analyte in pharmaceutical formulations by spectrophotometry and chromatography techniques.
These Secondary Standards are qualified as Certified Reference Materials. These are suitable for use in several analytical applications including but not limited to pharma release testing, pharma method development for qualitative and quantitative analyses, food and beverage quality control testing, and other calibration requirements.
分析報告
These secondary standards offer multi-traceability to the USP, EP (PhEur) and BP primary standards, where they are available.
其他說明
This Certified Reference Material (CRM) is produced and certified in accordance with ISO 17034 and ISO/IEC 17025. All information regarding the use of this CRM can be found on the certificate of analysis.
腳註
To see an example of a Certificate of Analysis for this material enter LRAA8969 in the slot below. This is an example certificate only and may not be the lot that you receive.
相關產品
产品编号
说明
价格
其他客户在看
Jörg D Leuppi et al.
JAMA, 309(21), 2223-2231 (2013-05-23)
International guidelines advocate a 7- to 14-day course of systemic glucocorticoid therapy in acute exacerbations of chronic obstructive pulmonary disease (COPD). However, the optimal dose and duration are unknown. To investigate whether a short-term (5 days) systemic glucocorticoid treatment in
Ian F Tannock et al.
The Lancet. Oncology, 14(8), 760-768 (2013-06-08)
Docetaxel plus prednisone is standard first-line chemotherapy for men with metastatic castrate-resistant prostate cancer. Aflibercept is a recombinant human fusion protein that binds A and B isoforms of VEGF and placental growth factor, thereby inhibiting angiogenesis. We assessed whether the
O Teuffel et al.
Leukemia, 25(8), 1232-1238 (2011-04-30)
This systematic review and meta-analysis compared the efficacy and toxicity of dexamethasone (DEX) versus prednisone (PRED) for induction therapy in childhood acute lymphoblastic leukemia (ALL). We searched biomedical literature databases and conference proceedings for randomized controlled trials comparing DEX and
M Dror Michaelson et al.
Journal of clinical oncology : official journal of the American Society of Clinical Oncology, 32(2), 76-82 (2013-12-11)
We evaluated angiogenesis-targeted sunitinib therapy in a randomized, double-blind trial of metastatic castration-resistant prostate cancer (mCRPC). Men with progressive mCRPC after docetaxel-based chemotherapy were randomly assigned 2:1 to receive sunitinib 37.5 mg/d continuously or placebo. Patients also received oral prednisone
Ronald de Wit
European journal of cancer (Oxford, England : 1990), 41(4), 502-507 (2005-03-02)
Until now, the use of systemic chemotherapy for advanced androgen-independent prostate cancer has had very little to offer to patients. However, in 2004, two large randomised trials investigating docetaxel vs. mitoxantrone have both demonstrated survival improvements, and, in one of
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系技术服务部门