422175
L-165,041
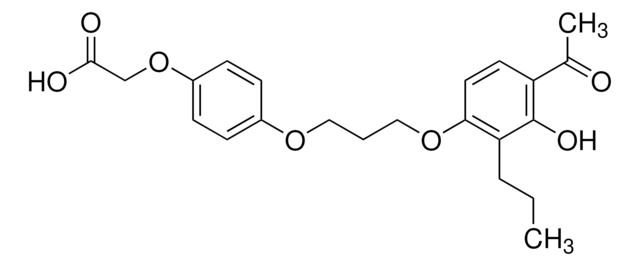
A cell-permeable phenoxyacetic acid derivative that acts as a potent and selective peroxisome proliferator activator receptor δ (PPARδ) agonist (Ki = 6 nM for hPPARδ and 730 nM for hPPARγ).
别名:
L-165,041, Compound P, L165041, 4-[3-(2-Propyl-3-hydroxy-4-acetyl)phenoxy]propyloxyphenoxy-acetic acid, PPAR Agonist VIII, PPARα Agonist III, PPARγ Agonist VII
登录查看公司和协议定价
所有图片(1)
About This Item
推荐产品
品質等級
化驗
≥98% (HPLC)
形狀
solid
製造商/商標名
Calbiochem®
儲存條件
OK to freeze
protect from light
顏色
white
溶解度
1 M NaOH: 40 mg/mL
DMSO: 5 mg/mL
運輸包裝
ambient
儲存溫度
2-8°C
InChI
1S/C22H26O7/c1-3-5-19-20(11-10-18(15(2)23)22(19)26)28-13-4-12-27-16-6-8-17(9-7-16)29-14-21(24)25/h6-11,26H,3-5,12-14H2,1-2H3,(H,24,25)
InChI 密鑰
HBBVCKCCQCQCTJ-UHFFFAOYSA-N
一般說明
A cell-permeable phenoxyacetic acid derivative that acts as a potent and selective peroxisome proliferator activator receptor δ (PPARδ) agonist (Ki = 6 nM for hPPAR δ and 730 nM for hPPARγ). Induces adipocyte differentiation in NIH-PPARδ cells at 500 nM. Also reported to raise total cholesterol in insulin-resistant db/db mice without altering glucose or triglyceride levels. Shown to increase uncoupling protein 3 (UCP3) gene expression in L6 myotubes. Also shown to inhibit cytokine-induced expression of vascular cell adhesion molecule-1 (VCAM-1) and the secretion of monocyte chemotactic protein-1 (MCP-1) in Eahy926 cells.
A cell-permeable phenoxyacetic acid derivative that acts as a potent and selective peroxisome proliferator activator receptor δ (PPARδ) agonist (Ki = 6 nM for hPPARδ and 730 nM for hPPARγ). Potently induces adipocyte differentiation in NIH-PPARδ cells at 500 nM and raises total cholesterol in insulin resistant db/db mice without altering glucose or triglycerides levels. Increases UCP3 (uncoupling protein 3) gene expression in L6 myotubes. Inhibits cytokine-induced expression of VCAM-1 (vascular cell adhesion molecule-1) and the secretion of MCP-1 (monocyte chemotactic protein-1) in EAhy926 cells.
生化/生理作用
Cell permeable: yes
Primary Target
Peroxisome proliferator activator receptor δ (PPAR@delta;)
Peroxisome proliferator activator receptor δ (PPAR@delta;)
Product does not compete with ATP.
Reversible: no
Target Ki: 6 nM for hPPARδ and 730 nM for hPPARγ
包裝
Packaged under inert gas
警告
Toxicity: Irritant (B)
重構
Following reconstitution aliquot and freeze (-20°C). Stock solutions are stable for up to 3 months at -20°C.
其他說明
Rival, Y., et al. 2002. Eur. J. Pharmacol.435, 143.
Castrillo, A., et al. 2001. J. Biol. Chem.276, 34082.
Hansen, J.B., et al. 2001. J. Biol. Chem.276, 3175.
Son, C., et al. 2001. Endocrinology142, 4189.
Wilkie, N., et al. 2001. J. Neurochem.78, 1135.
Leibowitz, M.D., et al. 2000. FEBS Lett.473, 333.
Berger, J., et al. 1999. J. Biol. Chem.274, 6718.
Lim, H., et al. 1999. Genes Dev.13, 1561.
Castrillo, A., et al. 2001. J. Biol. Chem.276, 34082.
Hansen, J.B., et al. 2001. J. Biol. Chem.276, 3175.
Son, C., et al. 2001. Endocrinology142, 4189.
Wilkie, N., et al. 2001. J. Neurochem.78, 1135.
Leibowitz, M.D., et al. 2000. FEBS Lett.473, 333.
Berger, J., et al. 1999. J. Biol. Chem.274, 6718.
Lim, H., et al. 1999. Genes Dev.13, 1561.
法律資訊
CALBIOCHEM is a registered trademark of Merck KGaA, Darmstadt, Germany
儲存類別代碼
11 - Combustible Solids
水污染物質分類(WGK)
WGK 3
閃點(°F)
Not applicable
閃點(°C)
Not applicable
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系技术服务部门