推荐产品
一般說明
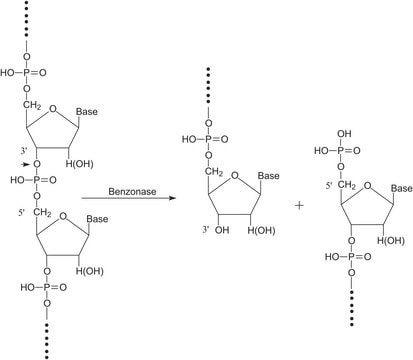
Benzonase® Nuclease is a genetically engineered endonuclease from Serratia marcescens. It degrades all forms of DNA and RNA (single stranded, double stranded, linear and circular) while having no proteolytic activity. It is effective over a wide range of conditions and possesses an exceptionally high specific activity. The enzyme completely digests nucleic acids to 5′-monophosphate terminated oligonucleotides (below the hybridization limit), which is ideal for removal of nucleic acids from recombinant proteins, enabling compliance with FDA guidelines for nucleic acid contamination. The ability of Benzonase to rapidly hydrolyze nucleic acids makes the enzyme an excellent choice for viscosity reduction to reduce processing time and increase yields of protein.
The enzyme consists of two subunits of 30 kDa each. It is functional between pH 6 and 10 and from 0-42°C and requires 1-2 mM Mg2+ for activation. The enzyme is also active in the presence of ionic and non-ionic detergents, reducing agents, PMSF (1 mM), EDTA (1 mM) and urea (relative activity depends on specific conditions). Activity is inhibited by <150 mM monovalent cations, <100 mM phosphate, <100 mM ammonium sulfate, or <100 mM guanidine HCl.
The enzyme consists of two subunits of 30 kDa each. It is functional between pH 6 and 10 and from 0-42°C and requires 1-2 mM Mg2+ for activation. The enzyme is also active in the presence of ionic and non-ionic detergents, reducing agents, PMSF (1 mM), EDTA (1 mM) and urea (relative activity depends on specific conditions). Activity is inhibited by <150 mM monovalent cations, <100 mM phosphate, <100 mM ammonium sulfate, or <100 mM guanidine HCl.
Benzonase® endonuclease Safety Plus Emprove® Expert is a companion product to our existing Benzonase® portfolio. All Benzonase® endonuclease products share the same amino acid sequence (proven by LC-MS/MS mass spectrometry), robustness and activity. Acting as endonuclease, degrading both DNA and RNA to small 3–5 base pairs (<6 kDa) fragments with no base preference, they are the ideal tool for nucleic acid removal in virus vector and vaccine manufacturing as demanded by the regulators. The use of Benzonase® endonuclease additionally increases the yield in virus purification, protects the downstream chromatography and filter devices from fouling and reduces feed stream viscosity. With its new features, Benzonase® endonuclease Safety Plus Emprove® Expert is ideally suited for manufacturing of cell and gene therapy agents such as adeno associated and lenti viruses, oncolytic viruses and viral vector vaccines.
Our SAFC® portfolio of high-quality products for biopharmaceutical processing withstands strict quality control procedures and is produced according to MQ-500 requirements as defined by the M-Clarity program.
M-Clarity Program
As part of our EMPROVE® Program, our raw materials are offered with EMPROVE® Dossiers which provide comprehensive, up-to-date documentation to help you navigate regulatory challenges, manage risks, and improve your manufacturing processes.
Our comprehensive portfolio of downstream process chemicals not only provides biopharmaceutical manufacturers with high-quality raw materials for production of classical and novel therapies, but also helps them get to market faster and simplify regulatory challenges. Ranging from non-GMP grades for low-risk application, to IPEC-PQG GMP for higher-risk applications, we have products covering all your manufacturing needs.
M-Clarity Program
As part of our EMPROVE® Program, our raw materials are offered with EMPROVE® Dossiers which provide comprehensive, up-to-date documentation to help you navigate regulatory challenges, manage risks, and improve your manufacturing processes.
Our comprehensive portfolio of downstream process chemicals not only provides biopharmaceutical manufacturers with high-quality raw materials for production of classical and novel therapies, but also helps them get to market faster and simplify regulatory challenges. Ranging from non-GMP grades for low-risk application, to IPEC-PQG GMP for higher-risk applications, we have products covering all your manufacturing needs.
應用
用于去除蛋白样品中的核酸。
DNA clearance for purification of biopharmaceuticals, viral vaccines, viral vector vaccines, viral vectros for cell and gene therapy applications as well as oncolytic viruses.
生化/生理作用
消化天然或热变性的DNA和RNA。
包裝
- 100,000 Units
- 500,000 Units
- 5,000,000 Units + 50,000 Units tailgate sample
準備報告
Benzonase® endonuclease Safety Plus Emprove® Expert is manufactured under good manufacturing practices (GMP ICH Q7)
法律資訊
Benzonase is a registered trademark of Merck KGaA, Darmstadt, Germany
Emprove is a registered trademark of Merck KGaA, Darmstadt, Germany
SAFC is a registered trademark of Merck KGaA, Darmstadt, Germany
儲存類別代碼
10 - Combustible liquids
水污染物質分類(WGK)
WGK 1
商品
Whether you are developing cell and gene therapies or viral vaccines, removing contaminating nucleic acids is an important part of viral production. The simplest way to do this is to use the Benzonase® endonuclease.
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系技术服务部门