About This Item
Recommended Products
biological source
synthetic (organic)
assay
≥99%
form
powder
solubility
1 M NaOH: soluble 50 mg/mL, clear to slightly hazy
NH4OH: freely soluble
NaOH: freely soluble
SMILES string
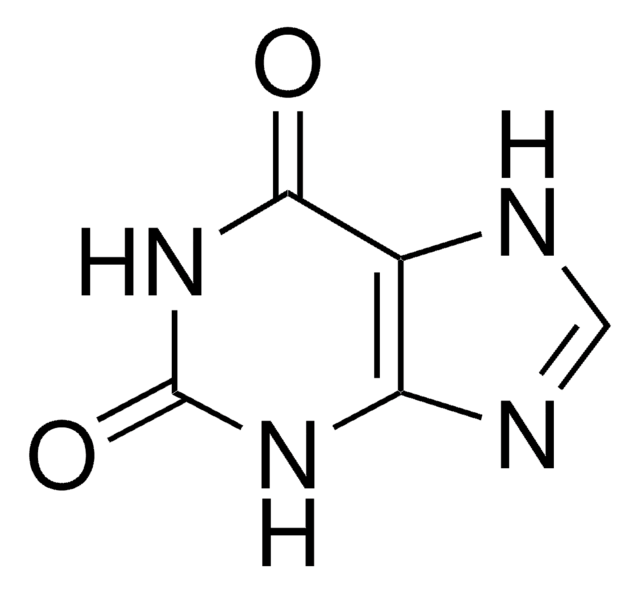
O=C1NC(=O)c2nc[nH]c2N1
InChI
1S/C5H4N4O2/c10-4-2-3(7-1-6-2)8-5(11)9-4/h1H,(H3,6,7,8,9,10,11)
InChI key
LRFVTYWOQMYALW-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
Application
Biochem/physiol Actions
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
ppe
dust mask type N95 (US), Eyeshields, Faceshields, Gloves
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Articles
Uric acid is not a direct additive to any of the classical cell culture media. However, it is present in animal sera. Consequently, uric acid is typically present in complete serum-supplemented classical cell culture systems.
Xanthine is a purine base found in most human body tissues and fluids as well as in other organisms. Methylated xanthines (methylxanthines), which include caffeine, paraxanthine, theobromine, and theophylline, commonly used for their effects as mild stiµlants and as bronchodilators, notably in the treatment of asthma symptoms. This application shows the efficient separation of several common xanthines and may be applied their analysis in any number of desired matrices.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service