G111080
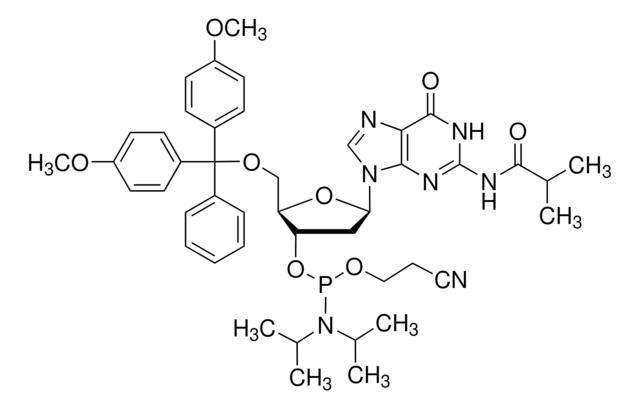
DMT-dG(ib) Phosphoramidite
configured for PerkinElmer, configured for Polygen
Synonym(s):
DMT-dG(ib) amidite, Guanosine, 5′-O-[bis(4-methoxyphenyl)phenylmethyl]-2′-deoxy-N-(2-methyl-1-oxopropyl)-, 3′-[2-cyanoethyl N,N-bis(1-methylethyl)phosphoramidite], N-(2-methyl-1-oxopropyl)-5′-O-[bis(4-methoxyphenyl)phenylmethyl]-2′-deoxyguanosine, 3′-[2-cyanoethyl N,N-bis(1-methylethyl)phosphoramidite], N2-Isobutyryl-5′-O-(4,4′-dimethoxytrityl)-2′-deoxyguanosine-3′-O-[O-(2-cyanoethyl)-N,N′-diisopropylphosphoramidite]
About This Item
Recommended Products
biological source
non-animal source (no BSE/TSE risk)
type
for DNA synthesis
product line
Proligo Reagents
assay
≥99% (31P-NMR)
≥99.0% (reversed phase HPLC)
form
powder or granules
technique(s)
oligo synthesis: suitable
impurities
≤0.3 wt. % water content (Karl Fischer)
color
white to off-white
λ
conforms (UV/VIS Identity)
compatibility
configured for PerkinElmer
configured for Polygen
nucleoside profile
base: deoxyguanosine
base protecting group: isobutyryl
2' protecting group: none
5' protecting group: DMT
deprotection: standard
storage temp.
2-8°C
SMILES string
COc1ccc(cc1)C(OC[C@H]2O[C@H](C[C@@H]2OP(OCCC#N)N(C(C)C)C(C)C)n3cnc4C(=O)NC(NC(=O)C(C)C)=Nc34)(c5ccccc5)c6ccc(OC)cc6
InChI
1S/C44H54N7O8P/c1-28(2)41(52)48-43-47-40-39(42(53)49-43)46-27-50(40)38-25-36(59-60(57-24-12-23-45)51(29(3)4)30(5)6)37(58-38)26-56-44(31-13-10-9-11-14-31,32-15-19-34(54-7)20-16-32)33-17-21-35(55-8)22-18-33/h9-11,13-22,27-30,36-38H,12,24-26H2,1-8H3,(H2,47,48,49,52,53)/t36-,37+,38+,60?/m0/s1
InChI key
FDRMKYVTIFSDPR-MMROLVBFSA-N
Looking for similar products? Visit Product Comparison Guide
General description
Key features of DNA phosphoramidites :
- Recommended cleavage and deprotection conditions are 8 hours at 55 °Cor 24 hours at room temperature using concentrated ammonia solution,for standard base-protected oligonucleotides
- The high coupling efficiency of Proligo′s DNA phosphoramidites leads tohigh-yield and high-quality oligonucleotidesThe product belongs to Expedite™ and PolyGen® synthesizers.
Legal Information
Storage Class
13 - Non Combustible Solids
wgk_germany
WGK 1
flash_point_f
Not applicable
flash_point_c
Not applicable
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service