A1806
Monoclonal Anti-Phosphotyrosine−Agarose antibody produced in mouse
clone PT-66, purified immunoglobulin, PBS solution
Synonym(s):
Monoclonal Anti-Phosphotyrosine, Phospho-Tyr, Phospho-tyrosine, p-Tyr
About This Item
Recommended Products
biological source
mouse
Quality Level
recombinant
expressed in mouse cell line
conjugate
agarose conjugate
antibody form
purified immunoglobulin
antibody product type
primary antibodies
clone
PT-66, monoclonal
form
PBS solution
technique(s)
immunoprecipitation (IP): suitable
isotype
IgG1
capacity
1 mg/mL binding capacity
shipped in
wet ice
storage temp.
2-8°C
target post-translational modification
phosphorylation (pTyr)
Looking for similar products? Visit Product Comparison Guide
Related Categories
General description
Immunogen
Application
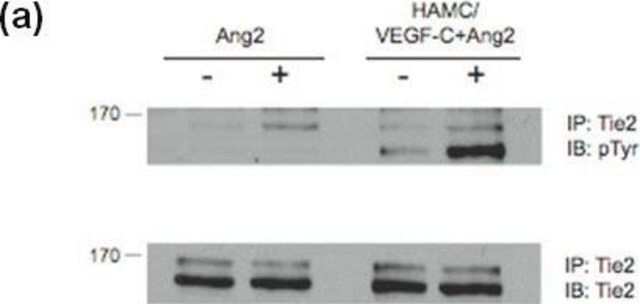
- immunoprecipitation experiments for affinity-purification of phosphotyrosine proteins
- phosphotyrosine pulldown assays
- PI3-kinase assay
Biochem/physiol Actions
Physical form
Disclaimer
Not finding the right product?
Try our Product Selector Tool.
Storage Class
10 - Combustible liquids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Articles
Post-translational modifications such as glycosylation, phosphorylation, and sulfation, to name a few, serve many functions. As a result, the analysis of proteins and their post-translational modifications is particularly important for the study of diseases where multiple genes are known to be involved, such as heart disease, cancer and diabetes.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service