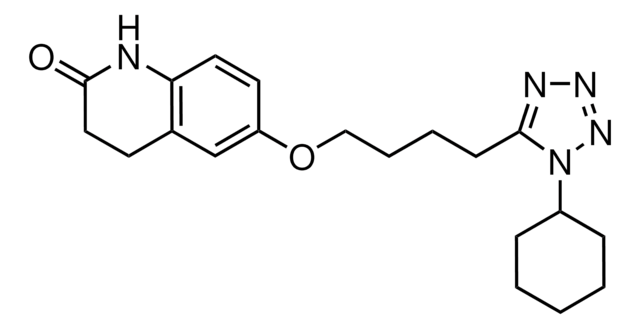
PHR1503
Cilostazol
Pharmaceutical Secondary Standard; Certified Reference Material
Synonym(s):
Cilostazol, 6-[4-(1-Cyclohexyl-1H-tetrazol-5-yl)-butoxy]-3,4-dihydro-2(1H)-quinolinone, OPC 13013, OPC 21, Pletaal
About This Item
Recommended Products
grade
certified reference material
pharmaceutical secondary standard
Quality Level
agency
traceable to USP 1134153
API family
cilostazol
CofA
current certificate can be downloaded
packaging
package of 1 g
technique(s)
HPLC: suitable
gas chromatography (GC): suitable
application(s)
pharmaceutical (small molecule)
format
neat
storage temp.
2-30°C
SMILES string
O=C1CCc2cc(OCCCCc3nnnn3C4CCCCC4)ccc2N1
InChI
1S/C20H27N5O2/c26-20-12-9-15-14-17(10-11-18(15)21-20)27-13-5-4-8-19-22-23-24-25(19)16-6-2-1-3-7-16/h10-11,14,16H,1-9,12-13H2,(H,21,26)
InChI key
RRGUKTPIGVIEKM-UHFFFAOYSA-N
Gene Information
human ... PDE3A(5139)
Looking for similar products? Visit Product Comparison Guide
General description
Application
Analysis Note
Other Notes
Footnote
Recommended products
Related product
signalword
Warning
hcodes
Hazard Classifications
Repr. 2
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
Choose from one of the most recent versions:
Certificates of Analysis (COA)
It looks like we've run into a problem, but you can still download Certificates of Analysis from our Documents section.
If you need assistance, please contact Customer Support.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service