AB2287
Anti-Amyloid Fibrils LOC Antibody
serum, Chemicon®
Synonym(s):
Amyloid Fibrils, Amyloid Fibrils LOC
About This Item
Recommended Products
biological source
rabbit
Quality Level
antibody form
serum
antibody product type
primary antibodies
clone
polyclonal
species reactivity
human
species reactivity (predicted by homology)
mouse, rat
manufacturer/tradename
Chemicon®
technique(s)
ELISA: suitable
dot blot: suitable
immunocytochemistry: suitable
immunohistochemistry: suitable
immunoprecipitation (IP): suitable
western blot: suitable
isotype
IgG
NCBI accession no.
UniProt accession no.
shipped in
wet ice
target post-translational modification
unmodified
Gene Information
human ... APP(351)
mouse ... App(11820)
General description
Specificity
Immunogen
Application
Quality
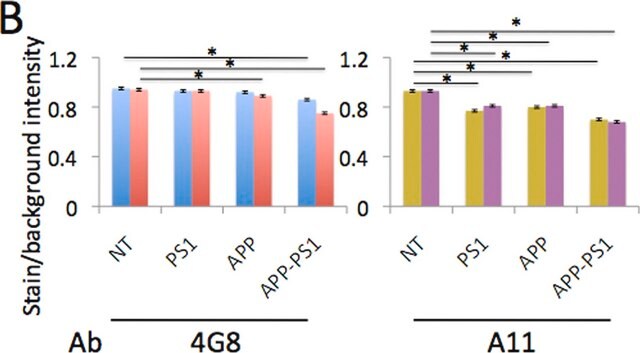
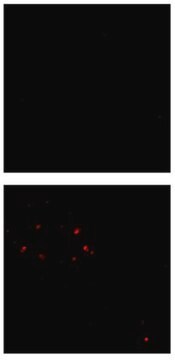
Dot Blot Analysis: 1:1,000 dilution of this antibody detected Amyloid fibrils in fibrils and monomers but not in prefibril oligos. A 1:5,000 dilution, as cited in Glabe C., et al. (2007) Mol Neurodegener 2, 18 shows that the binding with monomers is likely non-specific, and is a possible result of high primary antibody concentration.
Storage and Stability
Handling Recommendations: Upon receipt and prior to removing the cap, centrifuge the vial and gently mix the solution. Aliquot into microcentrifuge tubes and store at -20°C. Avoid repeated freeze/thaw cycles, which may damage IgG and affect product performance. After thawing, store at 4°C in 0.02% sodium azide.
Legal Information
Not finding the right product?
Try our Product Selector Tool.
Storage Class
10 - Combustible liquids
wgk_germany
WGK 1
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service