850400C
Avanti
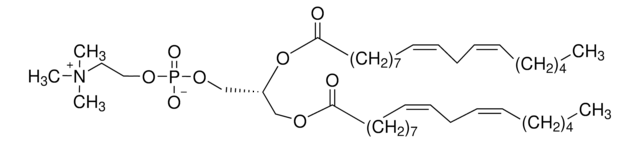
22:6 (Cis) PC
1,2-didocosahexaenoyl-sn-glycero-3-phosphocholine, chloroform
Synonym(s):
1,2-di-(4Z,7Z,10Z,13Z,16Z,19Z-docosahexaenoyl)-sn-glycero-3-phosphocholine; PC(22:6(4Z,7Z,10Z,13Z,16Z,19Z)/22:6(4Z,7Z,10Z,13Z,16Z,19Z))
About This Item
Recommended Products
assay
>99% (TLC)
form
liquid
packaging
pkg of 1 × 2.5 mL (850400C-25mg)
pkg of 5 × 4 mL (850400C-500mg)
manufacturer/tradename
Avanti Research™ - A Croda Brand 850400C
concentration
10 mg/mL (850400C-25mg)
25 mg/mL (850400C-500mg)
lipid type
cardiolipins
phospholipids
shipped in
dry ice
storage temp.
−20°C
InChI
1S/C52H80NO8P/c1-6-8-10-12-14-16-18-20-22-24-26-28-30-32-34-36-38-40-42-44-51(54)58-48-50(49-60-62(56,57)59-47-46-53(3,4)5)61-52(55)45-43-41-39-37-35-33-31-29-27-25-23-21-19-17-15-13-11-9-7-2/h8-11,14-17,20-23,26-29,32-35,38-41,50H,6-7,12-13,18-19,24-25,30-31,36-37,42-49H2,1-5H3/b10-8-,11-9-,16-14-,17-15-,22-20-,23-21-,28-26-,29-27-,34-32-,35-33-,40-38-,41-39-/t50-/m1/s1
InChI key
XLKQWAMTMYIQMG-SVUPRYTISA-N
General description
Application
- in liposomes, to study its effect on membrane vesiculation by dynamin and endophilin
- in multi-lamellar vesicles (MLVs) to analyze its effect on the biophysical properties of lipid membranes and on its interaction with a fragment of the Aβ peptide
- in lipid bilayers to study the influence of cholesterol on lateral segregation of saturated and unsaturated phospholipids
Packaging
Legal Information
signalword
Danger
Hazard Classifications
Acute Tox. 3 Inhalation - Acute Tox. 4 Oral - Aquatic Chronic 3 - Carc. 2 - Eye Irrit. 2 - Repr. 2 - Skin Irrit. 2 - STOT RE 1 - STOT SE 3
target_organs
Central nervous system, Liver,Kidney
Storage Class
6.1D - Non-combustible acute toxic Cat.3 / toxic hazardous materials or hazardous materials causing chronic effects
wgk_germany
WGK 3
flash_point_f
does not flash
flash_point_c
does not flash
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service