M79204
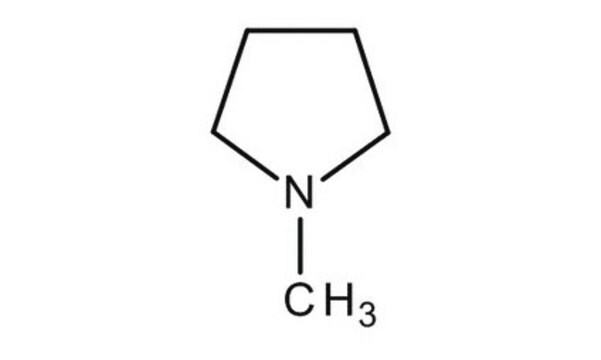
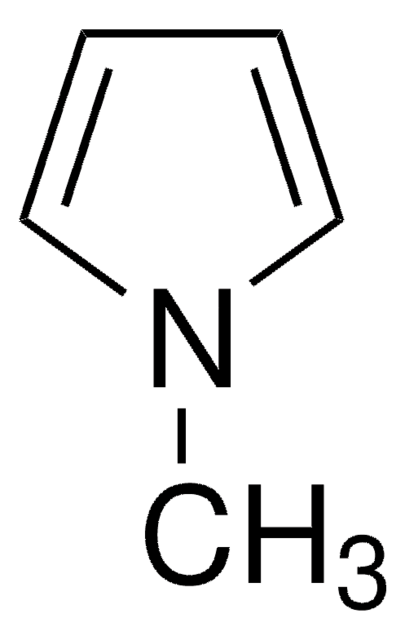
N-Methylpyrrolidine
97%
Synonym(s):
1-Methylpyrrolidine, N-Methylpyrrolidine, N-Methyltetrahydropyrrole
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Empirical Formula (Hill Notation):
C5H11N
CAS Number:
Molecular Weight:
85.15
Beilstein/REAXYS Number:
102445
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
assay
97%
form
liquid
bp
80-81 °C (lit.)
density
0.819 g/mL at 25 °C (lit.)
SMILES string
CN1CCCC1
InChI
1S/C5H11N/c1-6-4-2-3-5-6/h2-5H2,1H3
InChI key
AVFZOVWCLRSYKC-UHFFFAOYSA-N
Related Categories
Application
N-Methylpyrrolidine can be used as a starting material to synthesize:
N-methyl pyrrolidine-zinc borohydride (ZBHNMP), a reducing agent for reduction of aldehydes, ketones, acid chlorides, and esters. ZBHNMP is also used in the reductive amination of aldehydes and ketones to their corresponding amines.
N-alkyl-N-methyl-pyrrolidinium bis(trifluoromethanesulfonyl)imide ionic liquids.
N-methylpyrrolidine-2-one hydrotribromide applicable as a catalyst for aziridination of alkenes.
N-methyl pyrrolidine-zinc borohydride (ZBHNMP), a reducing agent for reduction of aldehydes, ketones, acid chlorides, and esters. ZBHNMP is also used in the reductive amination of aldehydes and ketones to their corresponding amines.
N-alkyl-N-methyl-pyrrolidinium bis(trifluoromethanesulfonyl)imide ionic liquids.
N-methylpyrrolidine-2-one hydrotribromide applicable as a catalyst for aziridination of alkenes.
signalword
Danger
hcodes
Hazard Classifications
Acute Tox. 3 Oral - Acute Tox. 4 Inhalation - Aquatic Chronic 2 - Flam. Liq. 2 - Skin Corr. 1A
Storage Class
3 - Flammable liquids
wgk_germany
WGK 3
flash_point_f
-0.4 °F - closed cup
flash_point_c
-18 °C - closed cup
ppe
Faceshields, Gloves, Goggles, type ABEK (EN14387) respirator filter
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Reductive amination of aldehydes and ketones to their corresponding amines with N-methylpyrrolidine zinc borohydride
Alinezhad H, et al.
Tetrahedron Letters, 50(6), 659-661 (2009)
N-methylpyrrolidine-2-one hydrotribromide: An efficient and new catalyst for the aziridination of alkenes using Chloramine-T under solvent free conditions
Jain SL, et al.
J. Mol. Catal. A: Chem., 256(1-2), 16-20 (2006)
Effect of the alkyl group on the synthesis and the electrochemical properties of N-alkyl-N-methyl-pyrrolidinium bis (trifluoromethanesulfonyl) imide ionic liquids
Appetecchi GB, et al.
Electrochimica Acta, 54(4), 1325-1332 (2009)
N-Methylpyrrolidine-zinc borohydride: As a new stable and efficient reducing agent in organic synthesis
Tajbakhsh M, et al.
Synthetic Communications, 33(2), 229-236 (2003)
Chafik Ghayor et al.
The Journal of biological chemistry, 286(27), 24458-24466 (2011-05-27)
Regulation of RANKL (receptor activator of nuclear factor κB ligand)-induced osteoclast differentiation is of current interest in the development of antiresorptive agents. Osteoclasts are multinucleated cells that play a crucial role in bone resorption. In this study, we investigated the
Global Trade Item Number
| SKU | GTIN |
|---|---|
| M79204-5ML | 4061834062493 |
| M79204-100ML | 4061834062479 |
| M79204-500ML | 4061834062486 |
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service