752452
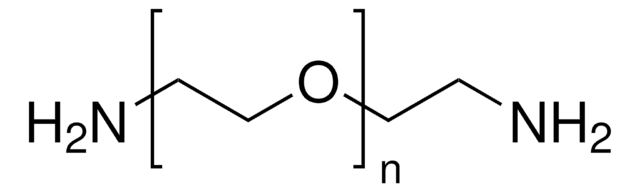
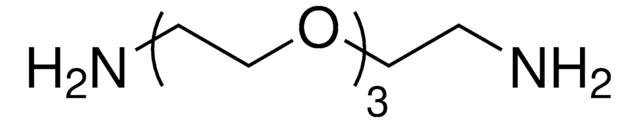
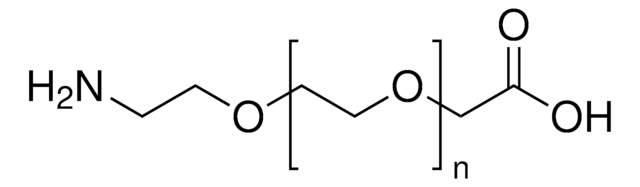
Poly(ethylene glycol) diamine
average MN 3,000, cross-linking reagent carboxyl reactive
Synonym(s):
Poly(ethylene glycol) bis(amine), Polyethylene glycol, O,O′-Bis(2-aminoethyl)polyethylene glycol, Diaminopolyethylene glycol, PEG-diamine, Polyoxyethylene bis(amine)
About This Item
Recommended Products
product name
Poly(ethylene glycol) diamine, average Mn 3,000
form
solid
Quality Level
mol wt
average Mn 3,000
reaction suitability
reagent type: cross-linking reagent
reactivity: carboxyl reactive
mp
55-60 °C
polymer architecture
shape: linear
functionality: homobifunctional
InChI
1S/C6H16N2O2/c7-1-3-9-5-6-10-4-2-8/h1-8H2
InChI key
IWBOPFCKHIJFMS-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Application
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
Choose from one of the most recent versions:
Certificates of Analysis (COA)
Don't see the Right Version?
If you require a particular version, you can look up a specific certificate by the Lot or Batch number.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
Highlighting new synthetic modifications of PEG to improve the mechanical properties and degradation of resulting hydrogels in tissue engineering applications.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service