666564
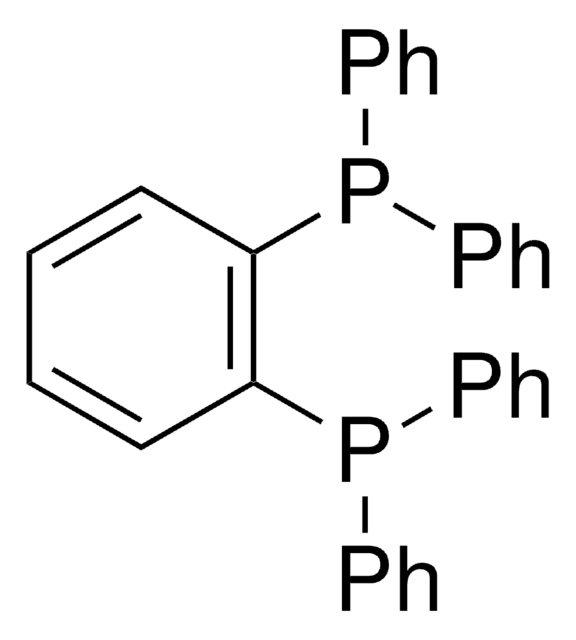
N-XantPhos
97%
Synonym(s):
NiXantphos, 4,6-Bis(diphenylphosphino)-10H-phenoxazine, 4,6-Bis(diphenylphosphino)phenoxazine
About This Item
Recommended Products
Quality Level
assay
97%
reaction suitability
reaction type: Buchwald-Hartwig Cross Coupling Reaction
reaction type: Heck Reaction
reaction type: Hiyama Coupling
reaction type: Negishi Coupling
reaction type: Sonogashira Coupling
reaction type: Stille Coupling
reaction type: Suzuki-Miyaura Coupling
reagent type: ligand
reaction type: Cross Couplings
mp
256-262 °C
functional group
phosphine
storage temp.
2-8°C
SMILES string
N1c2cccc(P(c3ccccc3)c4ccccc4)c2Oc5c1cccc5P(c6ccccc6)c7ccccc7
InChI
1S/C36H27NOP2/c1-5-15-27(16-6-1)39(28-17-7-2-8-18-28)33-25-13-23-31-35(33)38-36-32(37-31)24-14-26-34(36)40(29-19-9-3-10-20-29)30-21-11-4-12-22-30/h1-26,37H
InChI key
HSWZLYXRAOXOLL-UHFFFAOYSA-N
Application
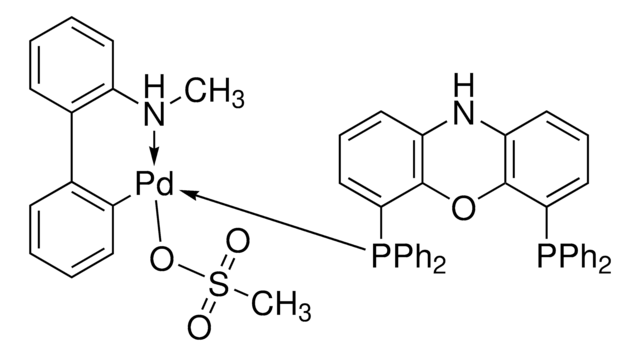
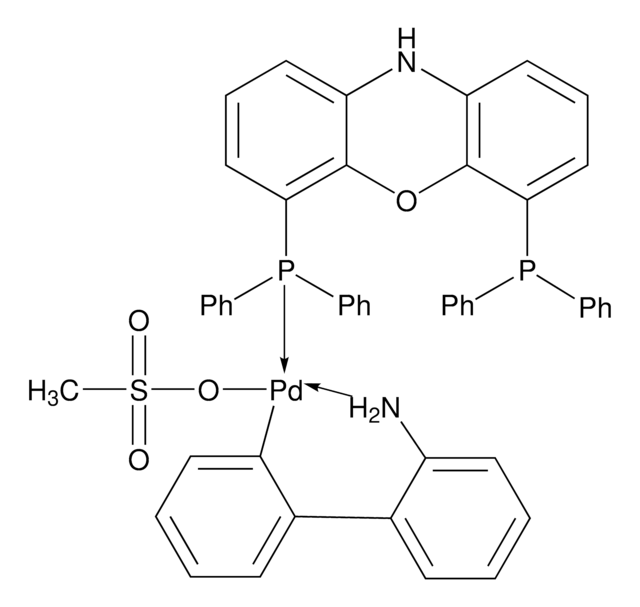
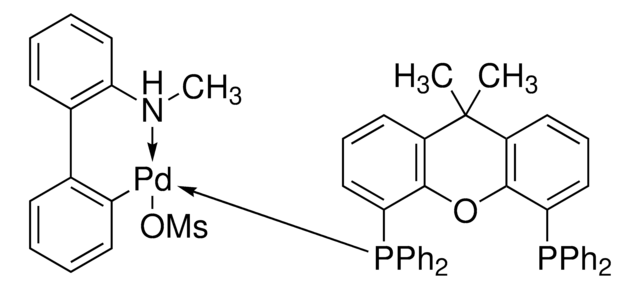
- The preparation of Pd-NiXantphos catalyst system for the room temperature cross-coupling reactions of unactivated aryl chlorides.
- The synthesis of cinchonine iridium(III) cyclometalated complex that exhibits luminescence and good quantum efficiency.
- N-XantPhos and N-modified counterparts are also used in the preparation of rhodium based catalysts for hydroformylation reaction.
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service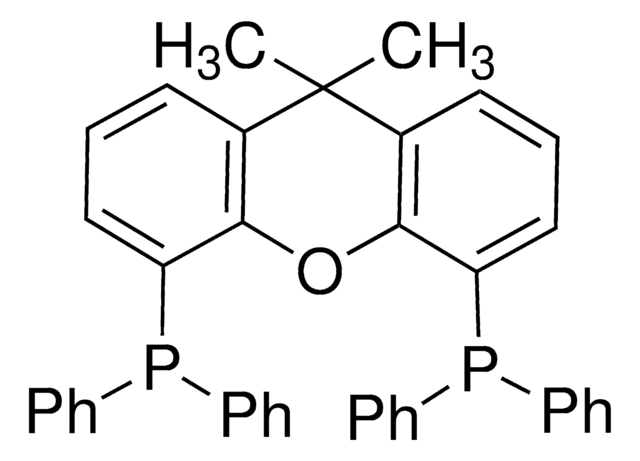




![Dichloro[9,9-dimethyl-4,5-bis(diphenylphosphino)xanthene]palladium(II) 95%](/deepweb/assets/sigmaaldrich/product/structures/374/597/f7932c5b-0448-498b-8254-f8ce1b9a4612/640/f7932c5b-0448-498b-8254-f8ce1b9a4612.png)