631493
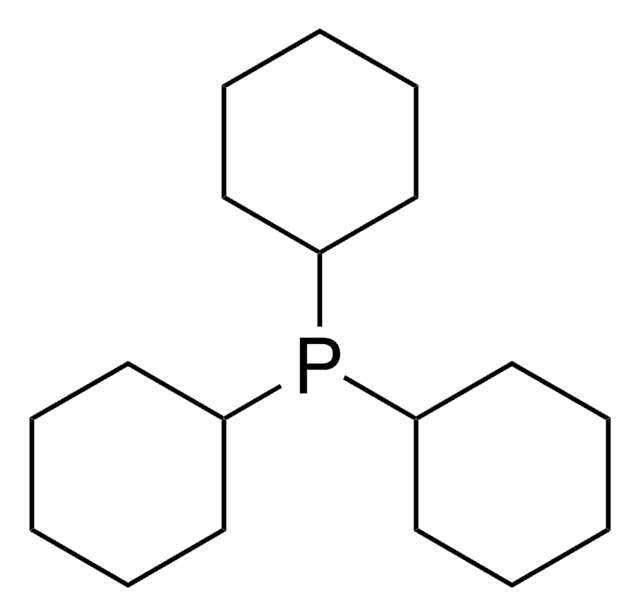
Tricyclohexylphosphine tetrafluoroborate
97%
Synonym(s):
Tricyclohexylphosphonium tetrafluoroborate
About This Item
Recommended Products
Quality Level
assay
97%
form
solid
reaction suitability
reaction type: Arylations
reaction type: Cross Couplings
reaction type: Sonogashira Coupling
reaction type: Stille Coupling
reaction type: Suzuki-Miyaura Coupling
mp
164 °C (lit.)
functional group
phosphine
SMILES string
F[B-](F)(F)F.[H][P+](C1CCCCC1)(C2CCCCC2)C3CCCCC3
InChI
1S/C18H33P.BF4/c1-4-10-16(11-5-1)19(17-12-6-2-7-13-17)18-14-8-3-9-15-18;2-1(3,4)5/h16-18H,1-15H2;/q;-1/p+1
InChI key
MYSMMEUXKHJYKH-UHFFFAOYSA-O
General description
Application
- As a ligand for preparing C-homoaporphine alkaloids via microwave-assisted direct-arylation.
- To synthesize poly-[9,9-bis(3-propylamide-2-methylpropyl sulfonic acid) fluorene]-co-(4,4′-diphenyl) (PFDBSO3H), which can be employed as a template and doping agent for enhancing the conductivity of poly(3,4-ethylenedioxythiophene) (PEDOT) films.
- To improve the reactivity of palladium-catalyzed Suzuki-Miyaura cross-coupling reaction between MIDA boronates and less activated alkenyl tosylates.
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Gloves, type N95 (US)
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
We are proud to offer a number of products used in catalytic amidation technology.
Global Trade Item Number
| SKU | GTIN |
|---|---|
| 631493-5G | 4061832721170 |
| 631493-1G | 4061826724392 |
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service

![[1,1′-Bis(diphenylphosphino)ferrocene]dichloropalladium(II)](/deepweb/assets/sigmaaldrich/product/structures/130/734/8846aa26-1858-458a-998d-8c306c13bf0f/640/8846aa26-1858-458a-998d-8c306c13bf0f.png)

![[1,1′-Bis(diphenylphosphino)ferrocene]dichloropalladium(II), complex with dichloromethane](/deepweb/assets/sigmaaldrich/product/structures/825/986/4317978b-1256-4c82-ab74-6a6a3ef948b1/640/4317978b-1256-4c82-ab74-6a6a3ef948b1.png)
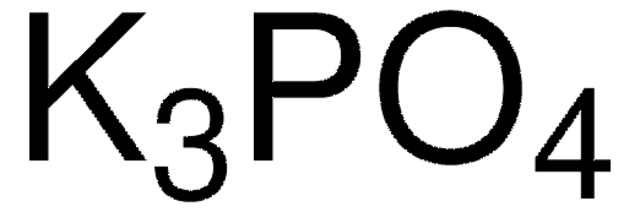
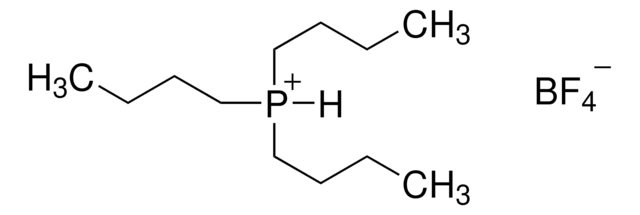
![[1,1′-Bis(di-cyclohexylphosphino)ferrocene]dichloropalladium(II) 98%](/deepweb/assets/sigmaaldrich/product/structures/136/854/a3142b2e-900c-47e5-8100-e48add9f4db6/640/a3142b2e-900c-47e5-8100-e48add9f4db6.png)