All Photos(3)
About This Item
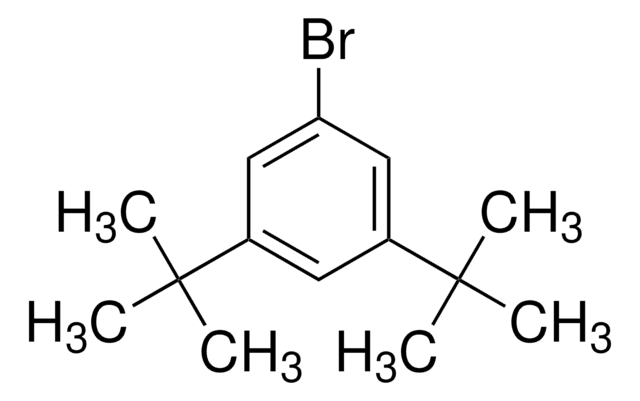
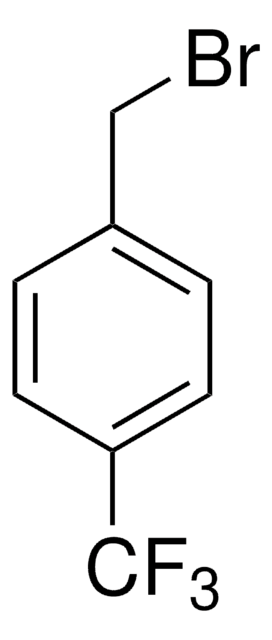
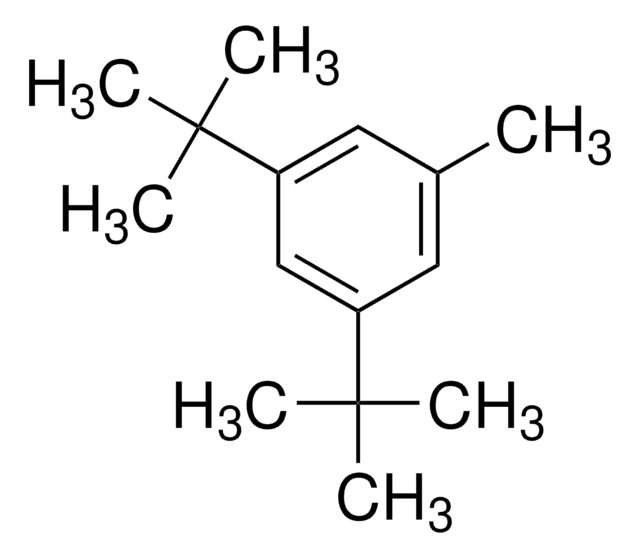
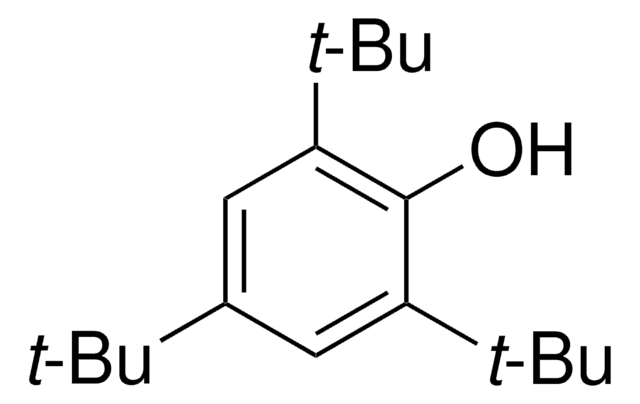
Linear Formula:
[(CH3)3C]3C6H2Br
CAS Number:
Molecular Weight:
325.33
Beilstein/REAXYS Number:
1913257
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
assay
97%
mp
168-173 °C (lit.)
functional group
bromo
SMILES string
CC(C)(C)c1cc(c(Br)c(c1)C(C)(C)C)C(C)(C)C
InChI
1S/C18H29Br/c1-16(2,3)12-10-13(17(4,5)6)15(19)14(11-12)18(7,8)9/h10-11H,1-9H3
InChI key
JOKZWHPYNRDCOA-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
1-Bromo-2,4,6-tri-tert-butylbenzene (2,4,6-tri-tert-butylbromobenzene) is a hindered aryl bromide. 1-Bromo-2,4,6-tri-tert-butylbenzene on reaction with phenylboronic acid yields α,α-dimethyl-β-phenyl hydrostyrene.
Application
1-Bromo-2,4,6-tri-tert-butylbenzene was used in the synthesis of bulky biarylphosphine ligand. This ligand was reported to participate in the Pd-catalyzed C-O cross-coupling of a wide range of aryl halides and phenols under milder conditions. It was used to investigate the effect on oligomerization of increased steric bulk in dimethylindium(III) chalcogenolates. It may be used to form α,α-dimethyl-β-phenyl hydrostyrene by reacting with phenylboronic acid.
signalword
Warning
hcodes
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
target_organs
Respiratory system
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
The first Cu-and amine-free Sonogashira-type cross-coupling in the C-6-alkynylation of protected 2'-deoxyadenosine.
Ngassa FN, et al.
Tetrahedron, 65(21), 4085-4091 (2009)
Luca Salvi et al.
Organic letters, 14(1), 170-173 (2011-12-21)
A new bulky biarylphosphine ligand (L8) has been developed that allows the Pd-catalyzed C-O cross-coupling of a wide range of aryl halides and phenols under milder conditions than previously possible. A direct correlation between the size of the ligand substituents
Palladium(0)-catalyzed intermolecular amination of unactivated C(sp³)-H bonds.
Jun Pan et al.
Angewandte Chemie (International ed. in English), 50(37), 8647-8651 (2011-08-04)
Glen G Briand et al.
Dalton transactions (Cambridge, England : 2003), 39(16), 3833-3841 (2010-04-08)
The effect on oligomerization of increased steric bulk in dimethylindium(III) chalcogenolates (Me(2)InER') (E = O, S, Se) has been examined. The facile reaction of Me(3)In with a series of phenols, thiophenols and selenophenols afforded the compounds [Me(2)InO(C(6)H(5))](2) (1), [Me(2)InO(2,6-Me(2)C(6)H(3))](2) (2)
Global Trade Item Number
| SKU | GTIN |
|---|---|
| 407097-5G | 4061832687810 |
| 407097-1G | 4061826002858 |
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service