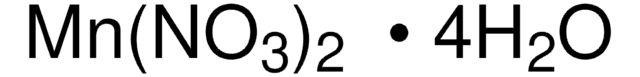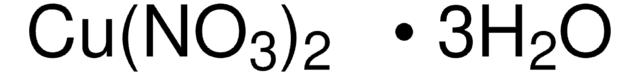288640
Manganese(II) nitrate hydrate
98%
Synonym(s):
Manganese(2+) dinitrate hydrate, Manganous dinitrate hydrate
About This Item
Recommended Products
Quality Level
assay
98%
form
solid
composition
Degree of hydration, 4-6
storage temp.
2-8°C
SMILES string
O.[Mn++].[O-][N+]([O-])=O.[O-][N+]([O-])=O
InChI
1S/Mn.2NO3.H2O/c;2*2-1(3)4;/h;;;1H2/q+2;2*-1;
InChI key
HBTFASPVVFSRRI-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
General description
Application
- A precursor for the synthesis of manganese dioxide (MnO2), which is a common cathode material in primary alkaline batteries.
- A precursor in the synthesis of manganese dioxide (MnO2) for use in flexible micro supercapacitors. MnO2 exhibits high specific capacitance and excellent cycling stability, making it suitable for rapid energy storage applications.
- A manganese source in the colloidal solution combustion synthesis process to produce 3D δ-MnO2 nanostructures with ultra-large mesopores used in the fabrication of high-performance lithium-ion battery anodes.
- A precursor in the microwave-aided fabrication of calcium-substituted dysprosium perovskite manganite oxide nanocomposites (DyMnO3) for supercapacitor applications.
signalword
Warning
hcodes
Hazard Classifications
Eye Irrit. 2 - Ox. Sol. 3 - Skin Irrit. 2 - STOT SE 3
target_organs
Respiratory system
Storage Class
5.1B - Oxidizing hazardous materials
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Gloves, type P3 (EN 143) respirator cartridges
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service