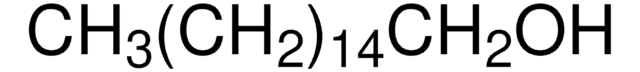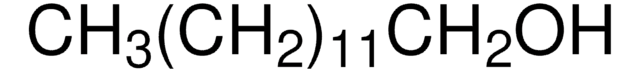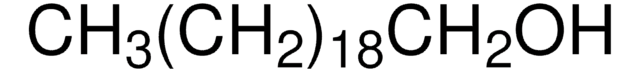258741
1-Hexadecanol
ReagentPlus®, 99%
Synonym(s):
Cetyl alcohol, Palmityl alcohol
About This Item
Recommended Products
grade
reagent
Quality Level
vapor density
8.34 (vs air)
vapor pressure
<0.01 mmHg ( 43 °C)
product line
ReagentPlus®
assay
99%
form
solid
autoignition temp.
483 °F
expl. lim.
8 %
dilution
(for general lab use)
bp
179-181 °C/10 mmHg (lit.)
mp
48-50 °C (lit.)
solubility
ethanol: soluble 100 mL/mL, clear to very slightly hazy, colorless
density
0.818 g/mL at 25 °C (lit.)
functional group
hydroxyl
SMILES string
CCCCCCCCCCCCCCCCO
InChI
1S/C16H34O/c1-2-3-4-5-6-7-8-9-10-11-12-13-14-15-16-17/h17H,2-16H2,1H3
InChI key
BXWNKGSJHAJOGX-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
General description
Application
- (±)-2-methoxyheptadecanoic acid (fatty acid)
- high-chain fatty acid esters of 1-hexadecanol, novel organic phase change material for thermal energy storage
- hexadecane (alkane) in the presence of membrane fraction of Vibrio furnissii M1
Legal Information
Storage Class
11 - Combustible Solids
wgk_germany
nwg
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Global Trade Item Number
| SKU | GTIN |
|---|---|
| 258741-1G | 4061826279342 |
| 258741-500G | 4061826062203 |
| 258741-100G | 4061826062142 |
| 258741-5G | 4061826062258 |
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service