All Photos(1)
About This Item
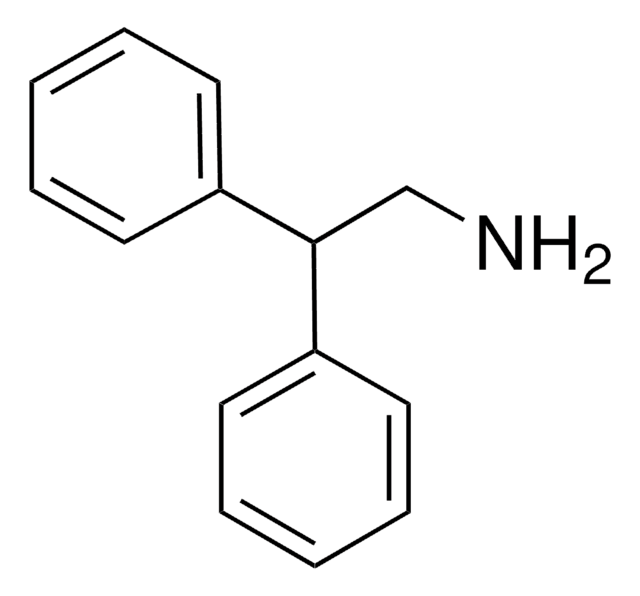
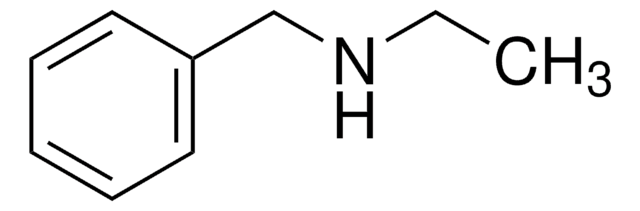
Linear Formula:
C6H5CH2CH(C6H5)NH2
CAS Number:
Molecular Weight:
197.28
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
product line
EMPROVE® Bio
assay
97%
form
liquid
refractive index
n20/D 1.58 (lit.)
bp
310-311 °C/750 mmHg (lit.)
density
1.02 g/mL at 25 °C (lit.)
SMILES string
NC(Cc1ccccc1)c2ccccc2
InChI
1S/C14H15N/c15-14(13-9-5-2-6-10-13)11-12-7-3-1-4-8-12/h1-10,14H,11,15H2
InChI key
DTGGNTMERRTPLR-UHFFFAOYSA-N
General description
(S)- and (R)-enantiomers of 1,2-diphenylethylamine are the precursors for synthesis of (S)- and (R)-1-(1,2-diphenylethyl)piperidine.
Legal Information
Emprove is a registered trademark of Merck KGaA, Darmstadt, Germany
signalword
Warning
hcodes
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
target_organs
Respiratory system
Storage Class
10 - Combustible liquids
wgk_germany
WGK 3
flash_point_f
235.4 °F - closed cup
flash_point_c
113 °C - closed cup
ppe
Eyeshields, Gloves, type ABEK (EN14387) respirator filter
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Michael L Berger et al.
Bioorganic & medicinal chemistry, 17(9), 3456-3462 (2009-04-07)
We resolved 1,2-diphenylethylamine (DPEA) into its (S)- and (R)-enantiomer and used them as precursors for synthesis of (S)- and (R)-1-(1,2-diphenylethyl)piperidine, flexible homeomorphs of the NMDA channel blocker MK-801. We also describe the synthesis of the dicyclohexyl analogues of DPEA. These
Carina S D Wink et al.
Analytical and bioanalytical chemistry, 407(6), 1545-1557 (2015-01-13)
Lefetamine (N,N-dimethyl-1,2-diphenylethylamine, L-SPA) was marketed as an opioid analgesic in Japan and Italy. After being widely abused, it became a controlled substance. It seems to be a pharmaceutical lead for designer drugs because N-ethyl-1,2-diphenylethylamine (NEDPA) and N-iso-propyl-1,2-diphenylethylamine (NPDPA) were confiscated
Daniel A Spudeit et al.
Journal of chromatography. A, 1363, 89-95 (2014-08-30)
This work reports a comparison of HPLC separations of enantiomers with chiral stationary phases (CSPs) prepared by chemically bonding cyclofructan-6, functionalized with isopropyl carbamate groups on fully and superficially porous particles (SPPs). The chromatographic performance of the superficially porous CSP
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service