Kluczowe dokumenty
T4951
2-((3-Trifluoromethyl)phenyl)histamine dimaleate
≥98% (HPLC), powder
Synonim(y):
(2-[2-(3-Trifluoromethyl)phenyl)-1H-imidazol-4-yl]ethanamine dimaleate, Compound 39
About This Item
Polecane produkty
Próba
≥98% (HPLC)
Formularz
powder
kolor
off-white
rozpuszczalność
H2O: ~20 mg/mL
temp. przechowywania
2-8°C
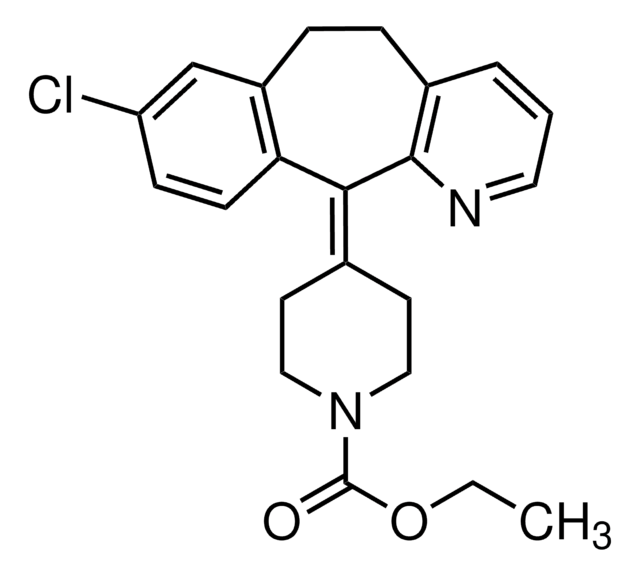
ciąg SMILES
OC(=O)\C=C/C(O)=O.OC(=O)\C=C/C(O)=O.NCCc1c[nH]c(n1)-c2cccc(c2)C(F)(F)F
InChI
1S/C12H12F3N3.2C4H4O4/c13-12(14,15)9-3-1-2-8(6-9)11-17-7-10(18-11)4-5-16;2*5-3(6)1-2-4(7)8/h1-3,6-7H,4-5,16H2,(H,17,18);2*1-2H,(H,5,6)(H,7,8)/b;2*2-1-
Klucz InChI
GGDUVPKRZGATLR-SPIKMXEPSA-N
Zastosowanie
Działania biochem./fizjol.
Cechy i korzyści
Uwaga dotycząca przygotowania
Hasło ostrzegawcze
Danger
Zwroty wskazujące rodzaj zagrożenia
Zwroty wskazujące środki ostrożności
Klasyfikacja zagrożeń
Acute Tox. 3 Oral - Eye Dam. 1 - Skin Irrit. 2 - STOT SE 3
Organy docelowe
Respiratory system
Kod klasy składowania
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
Klasa zagrożenia wodnego (WGK)
WGK 3
Temperatura zapłonu (°F)
Not applicable
Temperatura zapłonu (°C)
Not applicable
Środki ochrony indywidualnej
Eyeshields, Faceshields, Gloves, type P2 (EN 143) respirator cartridges
Wybierz jedną z najnowszych wersji:
Certyfikaty analizy (CoA)
Nie widzisz odpowiedniej wersji?
Jeśli potrzebujesz konkretnej wersji, możesz wyszukać konkretny certyfikat według numeru partii lub serii.
Masz już ten produkt?
Dokumenty związane z niedawno zakupionymi produktami zostały zamieszczone w Bibliotece dokumentów.
Nasz zespół naukowców ma doświadczenie we wszystkich obszarach badań, w tym w naukach przyrodniczych, materiałoznawstwie, syntezie chemicznej, chromatografii, analityce i wielu innych dziedzinach.
Skontaktuj się z zespołem ds. pomocy technicznej