Wszystkie zdjęcia(1)
Kluczowe dokumenty
M1508
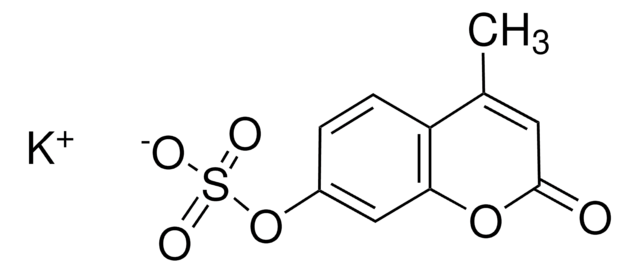
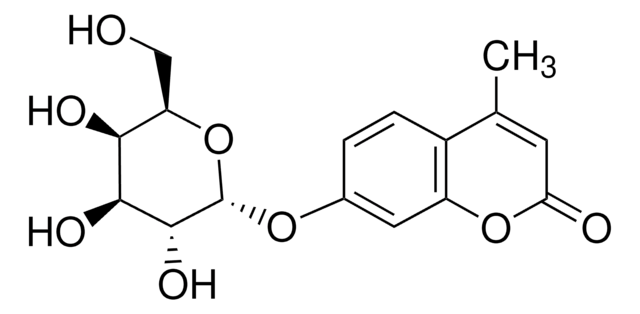
4-Methylumbelliferone sodium salt
fluorogenic, ≥98% (HPLC), crystalline
Synonim(y):
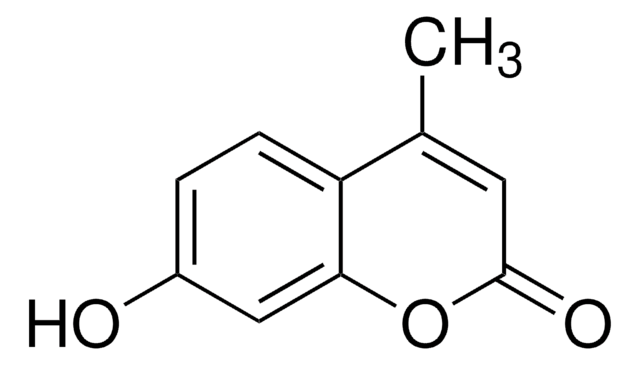
β-Methylumbelliferone, 7-Hydroxy-4-methylcoumarin
Zaloguj sięWyświetlanie cen organizacyjnych i kontraktowych
About This Item
Wzór liniowy:
C10H7O3Na
Numer CAS:
Masa cząsteczkowa:
198.15
Numer WE:
Numer MDL:
Kod UNSPSC:
12352204
Identyfikator substancji w PubChem:
NACRES:
NA.32
Polecane produkty
Nazwa produktu
4-Methylumbelliferone sodium salt, ≥98% (HPLC), crystalline
Próba
≥98% (HPLC)
Formularz
crystalline
kolor
yellow
rozpuszczalność
water: 50 mg/mL
ciąg SMILES
[Na+].CC1=CC(=O)Oc2cc([O-])ccc12
InChI
1S/C10H8O3.Na/c1-6-4-10(12)13-9-5-7(11)2-3-8(6)9;/h2-5,11H,1H3;/q;+1/p-1
Klucz InChI
JGMQHDNPUCPRQE-UHFFFAOYSA-M
Opis ogólny
4-methylumbelliferone (4-MU) is a coumarin derivative.
Zastosowanie
4-Methylumbelliferone sodium salt has been used:
- as a substrate to analyze the enzyme kinetics of its glucuronidation in the human liver
- as a standard to quantify the free 4-methylumbelliferone released as a result of enzyme-substrate action
- in cell survival assay to inhibit hyaluronan (HA) production and to study its effect on chemoresistance in ovarian cancer
Działania biochem./fizjol.
4-methylumbelliferone (4-MU) serves as a hyaluronan (HA) synthesis blocker. It exhibits anti-inflammatory, anti-fibrogenic, and antitumor properties. 4-MU is used to treat biliary spasms and might also be beneficial in nonalcoholic steatohepatitis (NASH), inflammation, hepatocyte injury, and fibrotic response. 4-MU is known to be glucuronidated by most of the uridine 5ฺ-diphospho-glucuronosyltransferase (UGT) isoforms of the human liver. This makes it a potent probe substrate to examine the overall hepatic UGTs activity.
Ta strona może zawierać tekst przetłumaczony maszynowo.
Kod klasy składowania
11 - Combustible Solids
Klasa zagrożenia wodnego (WGK)
WGK 3
Temperatura zapłonu (°F)
Not applicable
Temperatura zapłonu (°C)
Not applicable
Środki ochrony indywidualnej
Eyeshields, Gloves, type N95 (US)
Wybierz jedną z najnowszych wersji:
Masz już ten produkt?
Dokumenty związane z niedawno zakupionymi produktami zostały zamieszczone w Bibliotece dokumentów.
Klienci oglądali również te produkty
Valérie Jaulneau et al.
Journal of biomedicine & biotechnology, 2010, 525291-525291 (2010-05-07)
The industrial use of elicitors as alternative tools for disease control needs the identification of abundant sources of them. We report on an elicitor obtained from the green algae Ulva spp. A fraction containing most exclusively the sulfated polysaccharide known
Peter Pushko et al.
Virology, 501, 176-182 (2016-12-10)
Avian influenza (AI) viruses circulating in wild birds pose a serious threat to public health. Human and veterinary vaccines against AI subtypes are needed. Here we prepared triple-subtype VLPs that co-localized H5, H7 and H9 antigens derived from H5N1, H7N3
Rebecca M DuBois et al.
PLoS pathogens, 7(12), e1002398-e1002398 (2011-12-07)
Highly pathogenic avian influenza viruses of the H5N1 subtype continue to threaten agriculture and human health. Here, we use biochemistry and x-ray crystallography to reveal how amino-acid variations in the hemagglutinin (HA) protein contribute to the pathogenicity of H5N1 influenza
Ai-Sheng Xiong et al.
PloS one, 6(11), e26773-e26773 (2011-11-19)
A β-glucuronidase variant, GUS-TR3337, that was obtained by directed evolution exhibited higher thermostability than the wild-type enzyme, GUS-WT. In this study, the utility of GUS-TR337 as an improved reporter was evaluated. The corresponding gus-tr3337 and gus-wt genes were independently cloned
Gale E Smith et al.
Virology, 509, 90-97 (2017-06-19)
Avian influenza A (H5N1) viruses represent a growing threat for an influenza pandemic. The presence of widespread avian influenza virus infections further emphasizes the need for vaccine strategies for control of pre-pandemic H5N1 and other avian influenza subtypes. Influenza neuraminidase
Nasz zespół naukowców ma doświadczenie we wszystkich obszarach badań, w tym w naukach przyrodniczych, materiałoznawstwie, syntezie chemicznej, chromatografii, analityce i wielu innych dziedzinach.
Skontaktuj się z zespołem ds. pomocy technicznej