Kluczowe dokumenty
I4659
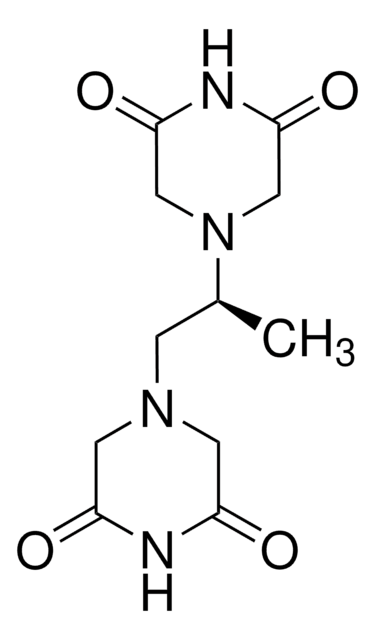
ICRF-193
apoptosis inducer, arabinosidase substrate
Synonim(y):
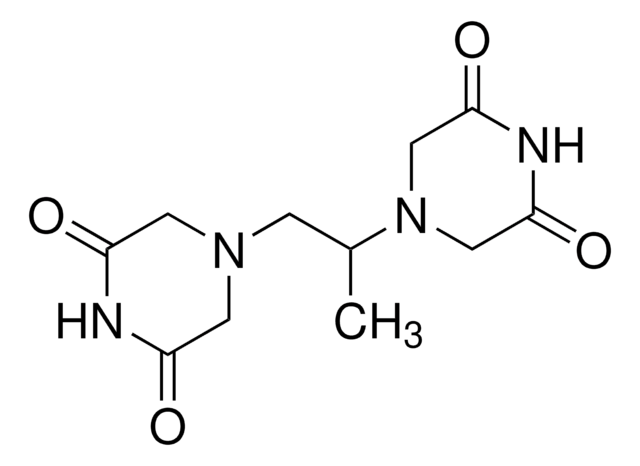
meso-4,4′-(3,2-Butanediyl)-bis(2,6-piperazinedione)
About This Item
Polecane produkty
pochodzenie biologiczne
synthetic (organic)
Poziom jakości
Próba
≥95%
Formularz
powder or flakes
solid
rozpuszczalność
DMSO: 4 mg/mL
temp. przechowywania
−20°C
ciąg SMILES
C[C@@H]([C@@H](C)N1CC(=O)NC(=O)C1)N2CC(=O)NC(=O)C2
InChI
1S/C12H18N4O4/c1-7(15-3-9(17)13-10(18)4-15)8(2)16-5-11(19)14-12(20)6-16/h7-8H,3-6H2,1-2H3,(H,13,17,18)(H,14,19,20)/t7-,8+
Klucz InChI
OBYGAPWKTPDTAS-OCAPTIKFSA-N
Opis ogólny
Zastosowanie
Działania biochem./fizjol.
Hasło ostrzegawcze
Danger
Zwroty wskazujące rodzaj zagrożenia
Zwroty wskazujące środki ostrożności
Klasyfikacja zagrożeń
Acute Tox. 3 Oral - Skin Sens. 1
Kod klasy składowania
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
Klasa zagrożenia wodnego (WGK)
WGK 3
Temperatura zapłonu (°F)
Not applicable
Temperatura zapłonu (°C)
Not applicable
Środki ochrony indywidualnej
dust mask type N95 (US), Eyeshields, Faceshields, Gloves
Wybierz jedną z najnowszych wersji:
Masz już ten produkt?
Dokumenty związane z niedawno zakupionymi produktami zostały zamieszczone w Bibliotece dokumentów.
Nasz zespół naukowców ma doświadczenie we wszystkich obszarach badań, w tym w naukach przyrodniczych, materiałoznawstwie, syntezie chemicznej, chromatografii, analityce i wielu innych dziedzinach.
Skontaktuj się z zespołem ds. pomocy technicznej