Wszystkie zdjęcia(6)
Kluczowe dokumenty
G4387
L-Glutamic Dehydrogenase (NADP) from Proteus sp.
buffered aqueous solution, ≥4,000 units/mL
Synonim(y):
L-Glutamate:NADP+ oxidoreductase (deaminating)
Zaloguj sięWyświetlanie cen organizacyjnych i kontraktowych
About This Item
Numer CAS:
Numer MDL:
Kod UNSPSC:
12352204
NACRES:
NA.54
Polecane produkty
pochodzenie biologiczne
bacterial (Proteus spp.)
Poziom jakości
Formularz
buffered aqueous solution
aktywność właściwa
≥4,000 units/mL
masa cząsteczkowa
~300 kDa
temp. przechowywania
2-8°C
Zastosowanie
This enzyme is useful for enzymatic determination of NH3, α-ketoglutaric acid and L-glutamic acid, and for assay of leucine aminopeptidase and urease. This enzyme is also used for enzymatic determination of urea when coupled with urease (URH-201) in clinical analysis. In vitro, various activity assays of this enzyme examine the conversion of α-ketoglutarate to L-glutamate, in the presence of excess ammonium ions (NH4+) and NADPH.
Działania biochem./fizjol.
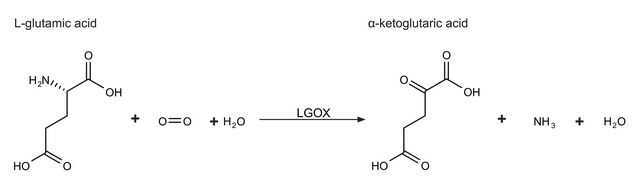
L-glutamic dehydrogenase catalyzes the conversion of glutamate to α-ketoglutarate.
Właściwości fizyczne
Isoelectric point : 4.6
Michaelis constants : 1.1 X 10-3M (NH3), 3.4 X 10-4M (α-Ketoglutarate)
1.2 X 10-3M (L-Glutamate), 1.4 X 10-5M (NADPH), 1.5 X 10-5M (NADP+)
Structure : 6 subunits (M.W.50,000) per mol of enzyme
Inhibitors : Hg++, Cd++, p-chloromercuribenzoate, pyridine, 4-4′-dithiopyridine,
2,2′-dithiopyridine
Optimum pH : 8.5 (α-KG→L-Glu) 9.8 (L-Glu→α-KG)
Optimum temperature : 45oC(α-KG−L-Glu) 45-55oC (L-Glu→α-KG)
pH stability : pH 6.0 - 8.5 (25oC, 20hr)
Thermal stability : below 50oC (pH 7.4, 10min)
Michaelis constants : 1.1 X 10-3M (NH3), 3.4 X 10-4M (α-Ketoglutarate)
1.2 X 10-3M (L-Glutamate), 1.4 X 10-5M (NADPH), 1.5 X 10-5M (NADP+)
Structure : 6 subunits (M.W.50,000) per mol of enzyme
Inhibitors : Hg++, Cd++, p-chloromercuribenzoate, pyridine, 4-4′-dithiopyridine,
2,2′-dithiopyridine
Optimum pH : 8.5 (α-KG→L-Glu) 9.8 (L-Glu→α-KG)
Optimum temperature : 45oC(α-KG−L-Glu) 45-55oC (L-Glu→α-KG)
pH stability : pH 6.0 - 8.5 (25oC, 20hr)
Thermal stability : below 50oC (pH 7.4, 10min)
Definicja jednostki
One unit will reduce 1.0 μmole of α-ketoglutarate to L-glutamate per min at pH 8.3 at 30 °C in the presence of ammonium ions and NADPH.
Postać fizyczna
Solution in 50 mM Tris HCl, pH 7.8, 5 mM Na2EDTA containing 0.05% sodium azide
Inne uwagi
Note: Do not confuse with non-specific L-GLDH, EC 1.4.1.3.
Ta strona może zawierać tekst przetłumaczony maszynowo.
Kod klasy składowania
10 - Combustible liquids
Klasa zagrożenia wodnego (WGK)
WGK 3
Temperatura zapłonu (°F)
Not applicable
Temperatura zapłonu (°C)
Not applicable
Wybierz jedną z najnowszych wersji:
Masz już ten produkt?
Dokumenty związane z niedawno zakupionymi produktami zostały zamieszczone w Bibliotece dokumentów.
Klienci oglądali również te produkty
J Bailey et al.
The Journal of biological chemistry, 257(10), 5579-5583 (1982-05-25)
The activity of bovine liver glutamate dehydrogenase is affected in several ways depending on substrate concentrations and pH. At ph 6.5 and below, both oxidative deamination and reductive amination reactions are inhibited by ADP. At pH 7.0 and above both
D P Hornby et al.
The Biochemical journal, 223(1), 161-168 (1984-10-01)
In steady-state kinetic studies of ox liver glutamate dehydrogenase in 0.11 M-potassium phosphate buffer, pH7, at 25 degrees C, the concentration of ADP was varied from 0.5 to 1000 microM. Inhibition was observed except when the concentrations of both glutamate
Daria V Borsakova et al.
Scientific reports, 12(1), 5437-5437 (2022-04-02)
Excessive ammonium blood concentration causes many serious neurological complications. The medications currently used are not very effective. To remove ammonium from the blood, erythrocyte-bioreactors containing enzymes that processing ammonium have been proposed. The most promising bioreactor contained co-encapsulated glutamate dehydrogenase
Tomomi Abiko et al.
Planta, 232(2), 299-311 (2010-05-06)
In plants, glutamine synthetase (GS) is the enzyme that is mainly responsible for the assimilation of ammonium. Conversely, in microorganisms such as bacteria and Ascomycota, NADP(H)-dependent glutamate dehydrogenase (GDH) and GS both have important roles in ammonium assimilation. Here, we
Daniel Juan Herrera et al.
Annals of clinical biochemistry, 47(Pt 1), 81-83 (2009-11-27)
Enzymatic assays using glutamate dehydrogenase (GLDH) to monitor the transformation of NAD(P)H to NAD(P)(+) by a spectrophotometric technique are the most common methods to measure plasma ammonia (PA) in routine laboratories worldwide. However, these assays can potentially be subject to
Nasz zespół naukowców ma doświadczenie we wszystkich obszarach badań, w tym w naukach przyrodniczych, materiałoznawstwie, syntezie chemicznej, chromatografii, analityce i wielu innych dziedzinach.
Skontaktuj się z zespołem ds. pomocy technicznej