375240
Antagonista HNF4, BI6015
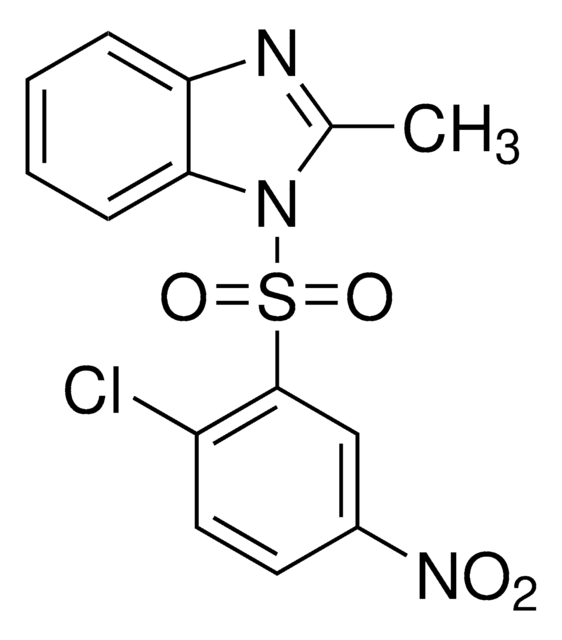
The HNF4 Antagonist, BI6, also referenced under CAS 93987-29-2, controls the biological activity of HNF4.
Synonim(y):
2-metylo-1-(2-metylo-5-nitrofenylosulfonylo)-1H-benzo[d]imidazol, antagonista czynnika jądrowego hepatocytów4
About This Item
Polecane produkty
Poziom jakości
Próba
≥99% (HPLC)
Formularz
powder
producent / nazwa handlowa
Calbiochem®
warunki przechowywania
OK to freeze
protect from light
kolor
beige
rozpuszczalność
DMSO: 10 mg/mL
Warunki transportu
ambient
temp. przechowywania
2-8°C
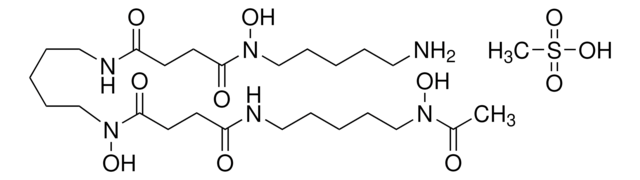
ciąg SMILES
CC1=NC2=C(N1S(C3=CC([N+]([O-])=O)=CC=C3C)(=O)=O)C=CC=C2
InChI
1S/C15H13N3O4S/c1-10-7-8-12(18(19)20)9-15(10)23(21,22)17-11(2)16-13-5-3-4-6-14(13)17/h3-9H,1-2H3
Klucz InChI
ILVCPQPMRPHZSG-UHFFFAOYSA-N
Opis ogólny
Działania biochem./fizjol.
HNFα i γ
Opakowanie
Ostrzeżenie
Inne uwagi
Informacje prawne
Kod klasy składowania
11 - Combustible Solids
Klasa zagrożenia wodnego (WGK)
WGK 2
Temperatura zapłonu (°F)
Not applicable
Temperatura zapłonu (°C)
Not applicable
Certyfikaty analizy (CoA)
Poszukaj Certyfikaty analizy (CoA), wpisując numer partii/serii produktów. Numery serii i partii można znaleźć na etykiecie produktu po słowach „seria” lub „partia”.
Masz już ten produkt?
Dokumenty związane z niedawno zakupionymi produktami zostały zamieszczone w Bibliotece dokumentów.
Nasz zespół naukowców ma doświadczenie we wszystkich obszarach badań, w tym w naukach przyrodniczych, materiałoznawstwie, syntezie chemicznej, chromatografii, analityce i wielu innych dziedzinach.
Skontaktuj się z zespołem ds. pomocy technicznej