T7168
Trypsin from porcine pancreas
tablet, 1 mg tablet
About This Item
Recommended Products
biological source
Porcine pancreas
Quality Level
form
tablet
specific activity
90-110% (compared to standard)
mol wt
23.8 kDa
storage temp.
−20°C
Looking for similar products? Visit Product Comparison Guide
Related Categories
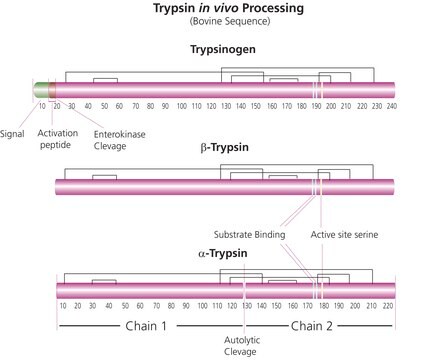
General description
Application
Biochem/physiol Actions
Serine protease inhibitors, including DFP, TLCK, APMSF, AEBSEF, and aprotinin, amongst others, will inhibit Trypsin.
Caution
Unit Definition
Preparation Note
For applications that require EDTA, solubilizing trypsin should be done with a buffered salt solution contaiing no Ca2+ or Mg2+.
inhibitor
substrate
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Resp. Sens. 1
Storage Class Code
11 - Combustible Solids
WGK
WGK 1
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Protocols
This procedure is for products with a specification for Trypsin activity using Na-Benzoyl-L-arginine ethyl ester (BAEE) as a substrate. The procedure is a continuous spectrophotometric rate determination (A253, Light path = 1 cm).
Use this protocol to for the entire immunohistochemistry (IHC) procedure through staining and visualization of specific antigens in paraffin-embedded tissue sections.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service