모든 사진(1)
About This Item
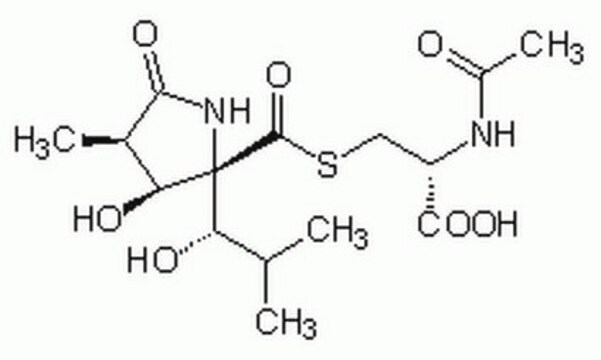
실험식(Hill 표기법):
C15H24N2O7S
CAS Number:
Molecular Weight:
376.43
MDL number:
UNSPSC 코드:
12352200
PubChem Substance ID:
NACRES:
NA.77
추천 제품
제품명
Lactacystin, ≥90% (HPLC)
Quality Level
분석
≥90% (HPLC)
양식
powder
효능
4 nM Ki (proteasome inhibitor)
solubility
water: 10 mg/mL, clear, colorless
저장 온도
−20°C
SMILES string
CC(C)[C@H](O)[C@]1(NC(=O)[C@H](C)[C@@H]1O)C(=O)SC[C@H](NC(C)=O)C(O)=O
InChI
1S/C15H24N2O7S/c1-6(2)10(19)15(11(20)7(3)12(21)17-15)14(24)25-5-9(13(22)23)16-8(4)18/h6-7,9-11,19-20H,5H2,1-4H3,(H,16,18)(H,17,21)(H,22,23)/t7-,9+,10+,11+,15-/m1/s1
InChI key
DAQAKHDKYAWHCG-RWTHQLGUSA-N
일반 설명
Lactacystin is an antibiotic and a metabolite of Streptomyces spp.
애플리케이션
Lactacystin has been used:
- as a proteasome inhibitor to inhibit protein degradation
- to inhibit proteasomal activity of cells for live cell imaging
- to block proteasomal proteolysis in human monocyte-derived dendritic cells (MoDCs) for 24 h
- to provide unilateral injection to animals to induce nigrostriatal lesions
생화학적/생리학적 작용
Lactacystin can block the development of cell cycle and stimulate differentiation in a murine neuroblastoma cell line. It can serve as a precursor for clasto-lactacystin β-lactone. Cell-permeable and irreversible proteasome inhibitor (Ki = 4nM). Inhibits NF-kB activation (IC50 = 10mM). Induces neurite outgrowth in neuro2A mouse neuroblastoma cells.
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
개인 보호 장비
Eyeshields, Gloves, type N95 (US)
이미 열람한 고객
Mechanistic Studies on the Inactivation of the Proteasome by Lactacystin A CENTRAL ROLE FOR clasto-LACTACYSTIN beta-LACTONE
Dick L R, et al.
The Journal of Biological Chemistry, 271(13), 7273-7276 (1996)
The development and pharmacology of proteasome inhibitors for the management and treatment of cancer
Ruggeri B, et al.
Advances in Pharmacology, 57(13), 91-135 (2009)
Cannabinoid receptor-induced neurite outgrowth is mediated by Rap1 activation through Galphao/i-triggered proteasomal degradation of Rap1GAPII
Jordan J D, et al.
The Journal of Biological Chemistry, 280(12), 11413-11421 (2005)
Evolution of extra-nigral damage predicts behavioural deficits in a rat proteasome inhibitor model of Parkinson's disease
Vernon A C, et al.
PLoS ONE, 6(2), e17269-e17269 (2011)
Dallas S Shi et al.
The Journal of clinical investigation, 124(9), 3757-3766 (2014-07-26)
The proteasome inhibiter bortezomib has been successfully used to treat patients with relapsed multiple myeloma; however, many of these patients become thrombocytopenic, and it is not clear how the proteasome influences platelet production. Here we determined that pharmacologic inhibition of
관련 콘텐츠
DISCOVER Bioactive Small Molecules for Nitric Oxide & Cell Stress Research
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.