추천 제품
형태
powder or crystals
Quality Level
색상
white to off-white
항생제 활성 스펙트럼
Gram-positive bacteria
fungi
동작 모드
cell membrane | interferes
cell wall synthesis | interferes
enzyme | inhibits
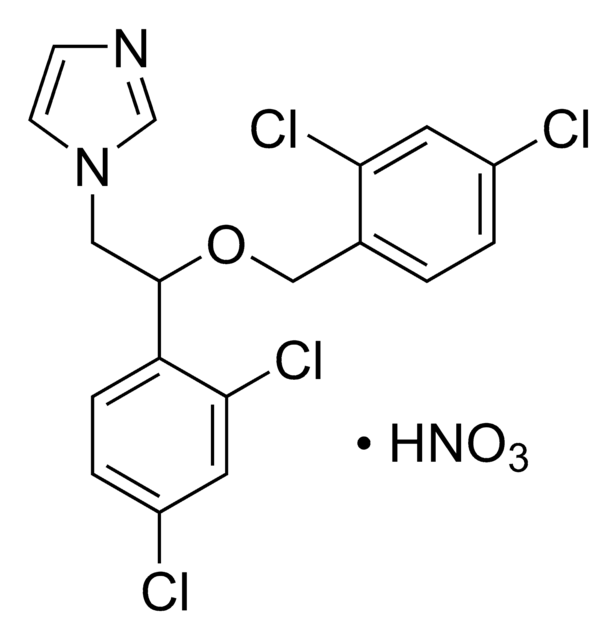
SMILES string
O[N+]([O-])=O.Clc1ccc(COC(Cn2ccnc2)c3ccc(Cl)cc3Cl)cc1
InChI
1S/C18H15Cl3N2O.HNO3/c19-14-3-1-13(2-4-14)11-24-18(10-23-8-7-22-12-23)16-6-5-15(20)9-17(16)21;2-1(3)4/h1-9,12,18H,10-11H2;(H,2,3,4)
InChI key
DDXORDQKGIZAME-UHFFFAOYSA-N
유사한 제품을 찾으십니까? 방문 제품 비교 안내
일반 설명
Chemical structure: imidazole
애플리케이션
Econazole is a broad spectrum antimycotic similar to ketoconazole. It has some action against Gram positive bacteria. It is used topically in dermatomycoses as well as orally and parenterally. It is used for studies on processes such as platelet serotonin uptake, prostanoid biosynthesis, EDHF-mediated relaxation, and Ca2+ transport pathways in thymic lymphocytes.
생화학적/생리학적 작용
Econazole interacts with 14-α demethylase, a cytochrome P-450 enzyme necessary to convert lanosterol to ergosterol. Inhibition of ergosterol results in increased cellular permeability causing leakage of cellular contents. Econazole may also inhibit endogenous respiration, interact with membrane phospholipids, inhibit the transformation of yeasts to mycelial forms, inhibit purine uptake, and impair triglyceride and phospholipid biosynthesis.
신호어
Warning
유해 및 위험 성명서
Hazard Classifications
Acute Tox. 4 Oral - Eye Irrit. 2 - Skin Irrit. 2
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
개인 보호 장비
dust mask type N95 (US), Eyeshields, Gloves
시험 성적서(COA)
제품의 로트/배치 번호를 입력하여 시험 성적서(COA)을 검색하십시오. 로트 및 배치 번호는 제품 라벨에 있는 ‘로트’ 또는 ‘배치’라는 용어 뒤에서 찾을 수 있습니다.
이미 열람한 고객
M A Ghannoum et al.
Clinical microbiology reviews, 12(4), 501-517 (1999-10-09)
The increased use of antibacterial and antifungal agents in recent years has resulted in the development of resistance to these drugs. The significant clinical implication of resistance has led to heightened interest in the study of antimicrobial resistance from different
Alessandro Malara et al.
The Journal of biological chemistry, 292(8), 3239-3251 (2017-01-05)
Abscisic acid (ABA) is a phytohormone involved in pivotal physiological functions in higher plants. Recently, ABA has been proven to be also secreted and active in mammals, where it stimulates the activity of innate immune cells, mesenchymal and hematopoietic stem
C J Jackson et al.
FEMS microbiology letters, 192(2), 159-162 (2000-11-07)
Azole antifungals are central to therapy and act by inhibiting a cytochrome P450, sterol 14-demethylase and blocking normal sterol synthesis. Our recent identification of a mycobacterial sterol biosynthetic pathway led us to probe the efficacy of a range of these
Inhibition of Ca2+ transport pathways in thymic lymphocytes by econazole, miconazole, and SKF 96365
M. J. Mason, B. Mayer, et al.
American Journal of Physiology. Cell Physiology, 264, C654-C662 (1993)
Núria Daranas et al.
Applied and environmental microbiology, 84(10) (2018-03-11)
A viability quantitative PCR (v-qPCR) assay was developed for the unambiguous detection and quantification of Lactobacillus plantarum PM411 viable cells in aerial plant surfaces. A 972-bp region of a PM411 predicted prophage with mosaic architecture enabled the identification of a
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.