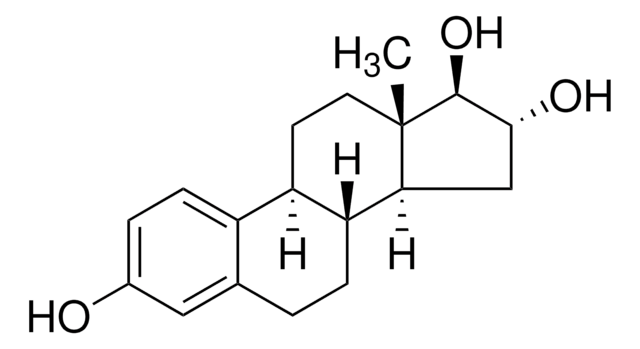
E1149
Estriol
meets USP testing specifications
동의어(들):
1,3,5(10)-Estratriene-3,16α,17β-triol, 16α-Hydroxyestradiol, 3,16α,17β-Trihydroxy-1,3,5(10)-estratriene
About This Item
추천 제품
Agency
USP/NF
meets USP testing specifications
mp
280-282 °C (lit.)
SMILES string
C[C@]12CC[C@H]3[C@@H](CCc4cc(O)ccc34)[C@@H]1C[C@@H](O)[C@@H]2O
InChI
1S/C18H24O3/c1-18-7-6-13-12-5-3-11(19)8-10(12)2-4-14(13)15(18)9-16(20)17(18)21/h3,5,8,13-17,19-21H,2,4,6-7,9H2,1H3/t13-,14-,15+,16-,17+,18+/m1/s1
InChI key
PROQIPRRNZUXQM-ZXXIGWHRSA-N
유전자 정보
human ... SERPINA6(866)
mouse ... Esr1(13982)
rat ... Ar(24208)
유사한 제품을 찾으십니까? 방문 제품 비교 안내
신호어
Danger
유해 및 위험 성명서
Hazard Classifications
Carc. 2 - Lact. - Repr. 1A
Storage Class Code
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
WGK
WGK 3
개인 보호 장비
Eyeshields, Gloves, type P3 (EN 143) respirator cartridges
시험 성적서(COA)
제품의 로트/배치 번호를 입력하여 시험 성적서(COA)을 검색하십시오. 로트 및 배치 번호는 제품 라벨에 있는 ‘로트’ 또는 ‘배치’라는 용어 뒤에서 찾을 수 있습니다.
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.