추천 제품
생물학적 소스
bacterial (Actinobacillus spp.)
양식
lyophilized powder
특이 활성도
≥4 units/mg solid
분자량
~100 kDa
저장 온도
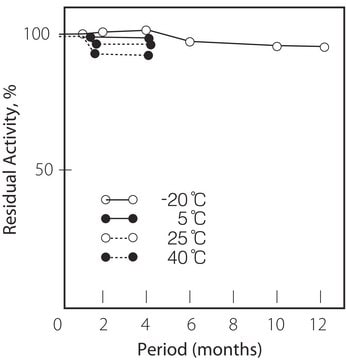
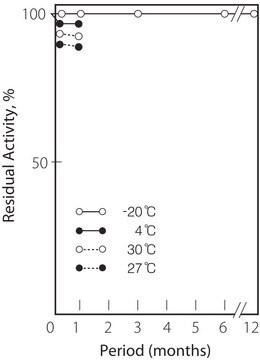
−20°C
일반 설명
Creatinase is a homodimer that catalyzes hydrolysis of creatine. It consists of two monomer subunits and two defined domains; N and C terminal domains. The C-terminal fold has both the α helices and anti-parallel β sheet within two structurally similar domains.In between these two domains, a sulfhydryl group acts as active site, and the activity is metal-independent.
애플리케이션
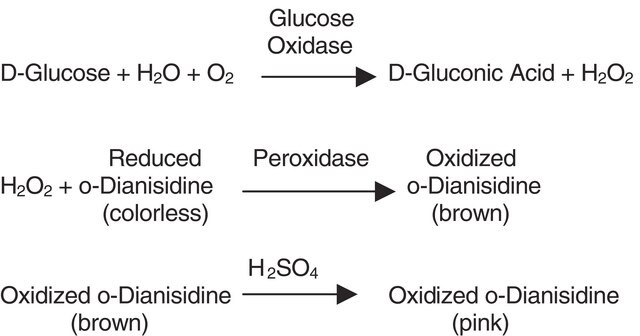
Creatinase mixed with sarcosine oxidase may be used to determine the level of creatine in different pH, temperature, enzyme ratio, and buffer concentration. It may also be used to determine the plasma creatinine level by using a centrifugal analyser.
생화학적/생리학적 작용
Creatinase accelerates the conversion reaction of creatine and water molecule to sarcosine and urea. It always acts in homodimer state and is induced by choline chloride.
물리적 특성
Isoelectric point: 4.6 ± 0.1
Michaelis constant: 1.9 x 10‾2M (Creatine)
Structure: 2 subunits per mole of enzyme
Inhibitors: Cu++, Hg++, Ag+
Optimum pH: 8.0
Optimum temp: 40°C
pH Stability: pH 5.5 − 9.0 (25°C, 16hr)
Thermal stability: Below 50°C (pH 7.5, 30 min)
Michaelis constant: 1.9 x 10‾2M (Creatine)
Structure: 2 subunits per mole of enzyme
Inhibitors: Cu++, Hg++, Ag+
Optimum pH: 8.0
Optimum temp: 40°C
pH Stability: pH 5.5 − 9.0 (25°C, 16hr)
Thermal stability: Below 50°C (pH 7.5, 30 min)
단위 정의
One unit will hydrolyze 1.0 μmole of creatine to urea and sarcosine per min at pH 7.5 at 37 °C.
물리적 형태
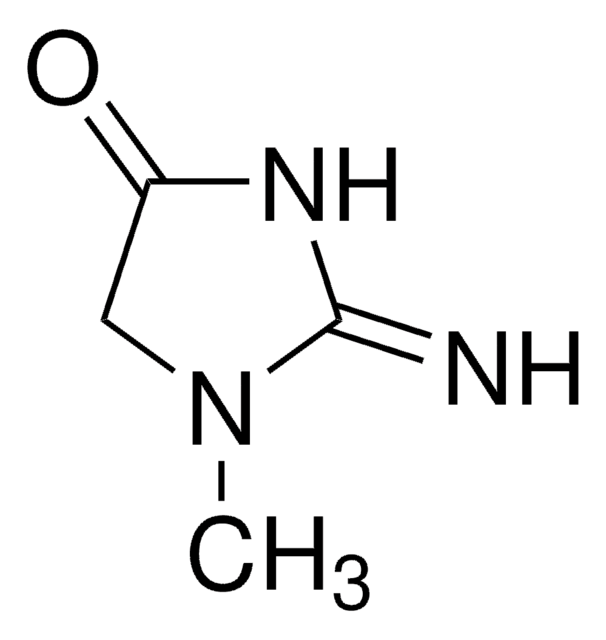
Lyophilized powder containing sugars and EDTA as stabilizers
신호어
Danger
유해 및 위험 성명서
예방조치 성명서
Hazard Classifications
Resp. Sens. 1
Storage Class Code
11 - Combustible Solids
WGK
WGK 1
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
개인 보호 장비
Eyeshields, Gloves, type N95 (US)
이미 열람한 고객
Balasundaram Padmanabhan et al.
Acta crystallographica. Section D, Biological crystallography, 58(Pt 8), 1322-1328 (2002-07-24)
The crystal structure of Actinobacillus creatine amidinohydrolase has been solved by molecular replacement. The amino-acid sequence has been derived from the crystal structure. Crystals belong to space group I222, with unit-cell parameters a = 111.26 (3), b = 113.62 (4)
H Crocker et al.
Journal of clinical pathology, 41(5), 576-581 (1988-05-01)
An enzymatic kit method for the determination of plasma creatinine was optimised for use with a centrifugal analyser and its performance characteristics and practicability compared with an end point and a kinetic Jaffé-based method. The enzymatic method exhibited several advantages
J F Bazan et al.
Proceedings of the National Academy of Sciences of the United States of America, 91(7), 2473-2477 (1994-03-29)
Amino acid sequence comparison suggests that the structure of Escherichia coli methionine aminopeptidase (EC 3.4.11.18) and the C-terminal domain of Pseudomonas putida creatinase (EC 3.5.3.3) are related. A detailed comparison of the three-dimensional folds of the two enzymes confirms this
Pinar Esra Erden et al.
Artificial cells, blood substitutes, and immobilization biotechnology, 34(2), 223-239 (2006-03-16)
A new enzyme electrode for the determination of creatine was developed by immobilizing creatinase (CI) and sarcosine oxidase (SO). The enzymes were co-immobilized in a poly(vinylferrocenium) matrix onto the surface of a platinum working electrode. Crosslinking with glutaraldehyte (GA) and
Jason A Berberich et al.
Acta biomaterialia, 1(2), 183-191 (2006-05-17)
The enzyme creatine amidinohydrolase is a clinically important enzyme used in the determination of creatinine in blood and urine. Continuous use biosensors are becoming more important in the clinical setting; however, long-use creatinine biosensors have not been commercialized due to
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.