23184
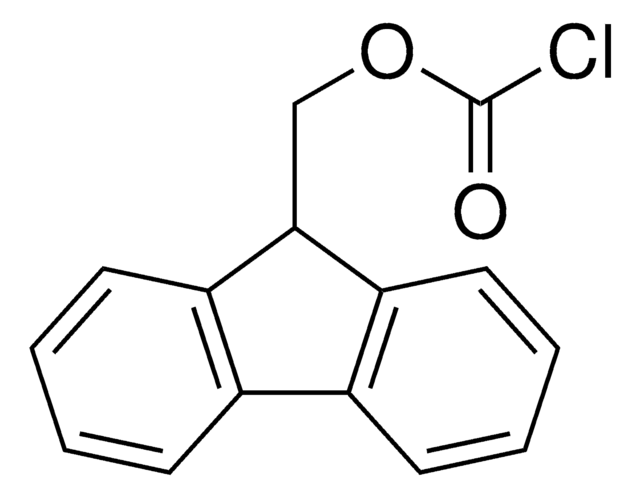
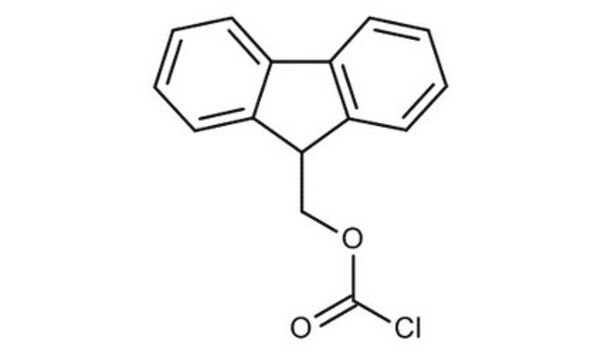
Fmoc chloride
BioReagent, ≥99.0% (HPLC)
동의어(들):
9-Fluorenylmethoxycarbonyl chloride, 9-Fluorenylmethyl chloroformate, Fmoc-Cl
로그인조직 및 계약 가격 보기
모든 사진(1)
About This Item
실험식(Hill 표기법):
C15H11ClO2
CAS Number:
Molecular Weight:
258.70
Beilstein:
2279177
EC Number:
MDL number:
UNSPSC 코드:
12352108
PubChem Substance ID:
NACRES:
NA.22
추천 제품
애플리케이션
9-Fluorenylmethoxycarbonyl chloride (Fmoc-Cl) is a reagent for pre-column derivatization of amino acids and biogenic amines for HPLC amino acid analysis and for preparing N-Fmoc amino acids for solid-phase peptide synthesis.
Reagent for derivatizing amino acids for HPLC amino acid analysis and for preparing N-Fmoc amino acids for solid-phase peptide synthesis.
기타 정보
Reagent for the pre-column fluorescent derivatization of amino acids
신호어
Danger
유해 및 위험 성명서
Hazard Classifications
Eye Dam. 1 - Skin Corr. 1B
보충제 위험성
Storage Class Code
8A - Combustible corrosive hazardous materials
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
개인 보호 장비
Eyeshields, Faceshields, Gloves, type P3 (EN 143) respirator cartridges
이미 열람한 고객
Valentin Lozanov et al.
Journal of chromatography. B, Analytical technologies in the biomedical and life sciences, 860(1), 92-97 (2007-11-07)
A liquid chromatography method for simultaneous analysis of amino acids, polyamines, catecholeamines and metanephrines in human body fluids after derivatization with 9-fluorenylmethyloxycarbonyl chloride was developed. The chromatographic behavior of analytes at different pH of mobile phase was studied. Successful baseline
Ibolya Molnár-Perl
Journal of chromatography. B, Analytical technologies in the biomedical and life sciences, 879(17-18), 1241-1269 (2011-03-05)
An overview is presented on the advancement of the two most frequently used derivatization protocols applying the o-phthalaldehyde (OPA)-thiol and the fluorenylmethyloxycarbonyl (FMOC) chloride reagents, prior to the high performance liquid chromatographic analysis of amino acids. This review pays special
E J Miller et al.
Analytical biochemistry, 190(1), 92-97 (1990-10-01)
A recently described procedure for amino acid analyses has been modified and adapted for use in quantitating the unique mixture of products commonly found in hydrolysates of the collagens. The method involves precolumn derivatization of hydrolysates with 9-fluorenylmethyl chloroformate (FMOC-CL)
A Jámbor et al.
Journal of chromatography. A, 1216(34), 6218-6223 (2009-07-28)
This paper, as a novelty to this field, presents the deproteinization and derivatization of plasma's free amino acids (PFAAs), simultaneously, in a single step, with the acetonitrile (ACN) containing 9-fluorenylmethyloxycarbonyl chloride (FMOC) reagent. Deproteinization and derivatization, were studied with 22
P Fürst et al.
Journal of chromatography, 499, 557-569 (1990-01-19)
Reversed-phase high-performance liquid chromatography (RP-HPLC) is a powerful method for assaying physiological amino acid concentrations in biological fluids. Four pre-column derivatization methods, with o-phthaldialdehyde (OPA), 9-fluorenylmethyl chloroformate (FMOC-Cl), phenyl isothiocyanate (PITC) and 1-dimethylaminonaphthalene-5-sulphonyl chloride (dansyl-Cl), were assessed with respect to
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.