모든 사진(1)
About This Item
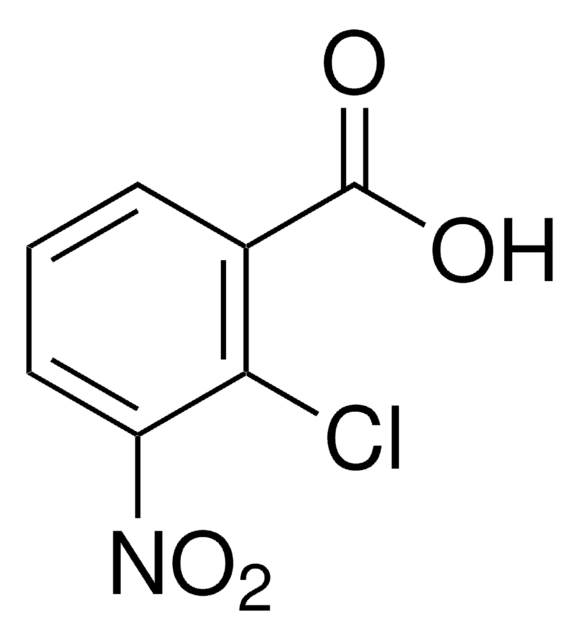
실험식(Hill 표기법):
C24H14
CAS Number:
Molecular Weight:
302.37
Beilstein:
2054067
MDL number:
UNSPSC 코드:
41116107
PubChem Substance ID:
NACRES:
NA.24
추천 제품
Grade
certified reference material
Agency
BCR®
제조업체/상표
JRC
기술
HPLC: suitable
gas chromatography (GC): suitable
형식
neat
저장 온도
2-8°C
SMILES string
c1ccc2c(c1)cc3ccc4c5ccccc5cc6ccc2c3c46
InChI
1S/C24H14/c1-3-7-19-15(5-1)13-17-9-12-22-20-8-4-2-6-16(20)14-18-10-11-21(19)23(17)24(18)22/h1-14H
InChI key
RXUSYFJGDZFVND-UHFFFAOYSA-N
유사한 제품을 찾으십니까? 방문 제품 비교 안내
분석 메모
For more information please see:
BCR159
BCR159
법적 정보
BCR is a registered trademark of European Commission
신호어
Danger
유해 및 위험 성명서
Hazard Classifications
Carc. 1B - Muta. 2
Storage Class Code
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
가장 최신 버전 중 하나를 선택하세요:
G A Marsch et al.
Chemical research in toxicology, 5(6), 765-772 (1992-11-01)
The in vivo formation of dibenzo[a,h]pyrene-DNA adducts in mouse skin was assessed by laser-excited fluorescence spectroscopy at 77 and 4.2 K. Two adducts were identified with fluorescence origin bands at approximately 383.5 and 407.2 nm, and these were shown to
Tumorigenicity of bay-region diol-epoxides and other benzo-ring derivatives of dibenzo(a,h)pyrene and dibenzo(a,i)pyrene on mouse skin and in newborn mice.
R L Chang et al.
Cancer research, 42(1), 25-29 (1982-01-01)
Dibenzo[a,h]pyrene.
IARC monographs on the evaluation of the carcinogenic risk of chemicals to humans, 32, 331-335 (1983-12-01)
A W Wood et al.
Cancer research, 41(7), 2589-2597 (1981-07-01)
The mutagenic activities of dibenzo(a,h)(pyrene, dibenzo(a,i)pyrene, and a total of 11 of their benzo-ring derivatives were evaluated in bacterial and mammalian cells in the absence or presence of a mammalian metabolic activation system. trans-1,2-Dihydroxy-1,2-dihydrodibenzo(a,h)pyrene and trans-3,4-dihydroxy-3,4-dihydrodibenzo(a,i)pyrene, the expected dihydrodiol precursors
B S Hass et al.
Cancer research, 42(5), 1646-1649 (1982-05-01)
The polycyclic aromatic hydrocarbons dibenzo(a,i)pyrene and dibenzo(a,h)pyrene, each of which possesses two bay regions, and their bay-region difluorinated derivatives were tested for mutagenicity for ouabain and 6-thioguanine resistance in Chinese hamster V79 cells. Since V79 cells do not metabolize polycyclic
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.![Dibenzo[a,l]pyrene BCR®, certified reference material](/deepweb/assets/sigmaaldrich/product/structures/937/930/3e2321b0-d54a-46c2-bb84-007bb57eb381/640/3e2321b0-d54a-46c2-bb84-007bb57eb381.png)
![Dibenzo[a,e]pyrene BCR®, certified reference material](/deepweb/assets/sigmaaldrich/product/structures/276/556/dd214831-9057-438a-b33d-aa10f7b4e48c/640/dd214831-9057-438a-b33d-aa10f7b4e48c.png)
![Benzo[a]pyrene ≥96% (HPLC)](/deepweb/assets/sigmaaldrich/product/structures/253/820/be96d879-1811-46c0-8f11-612019691c2d/640/be96d879-1811-46c0-8f11-612019691c2d.png)