모든 사진(1)
About This Item
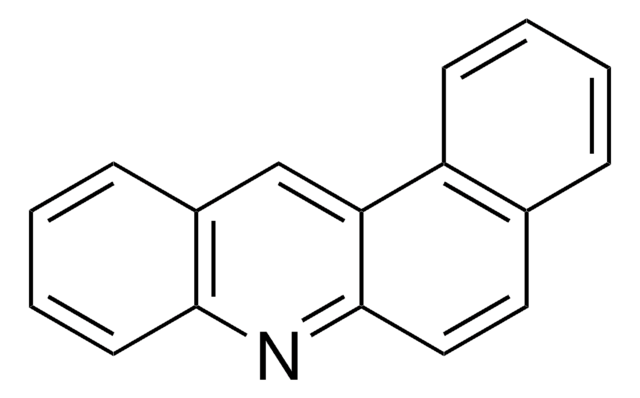
실험식(Hill 표기법):
C17H11N
CAS Number:
Molecular Weight:
229.28
Beilstein:
9262
MDL number:
UNSPSC 코드:
41116107
PubChem Substance ID:
NACRES:
NA.24
추천 제품
Grade
certified reference material
Agency
BCR®
제조업체/상표
JRC
기술
HPLC: suitable
gas chromatography (GC): suitable
형식
neat
저장 온도
2-8°C
SMILES string
c1ccc2nc3ccc4ccccc4c3cc2c1
InChI
1S/C17H11N/c1-3-7-14-12(5-1)9-10-17-15(14)11-13-6-2-4-8-16(13)18-17/h1-11H
InChI key
JEGZRTMZYUDVBF-UHFFFAOYSA-N
분석 메모
For more information please see:
BCR157
BCR157
법적 정보
BCR is a registered trademark of European Commission
신호어
Warning
유해 및 위험 성명서
예방조치 성명서
Hazard Classifications
Acute Tox. 4 Oral - Aquatic Acute 1 - Aquatic Chronic 1
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
가장 최신 버전 중 하나를 선택하세요:
R P Deutsch-Wenzel et al.
Cancer letters, 20(1), 97-101 (1983-08-01)
Using a beeswax/tricaprylin mixture as vehicle, three doses each of acridine, benz[a]acridine (BaAC), benz[c]acridine (BcAC), dibenz[a,h]-acridine (DBa,hAC) and dibenz[a,j]acridine (DBa,jAC) were injected into the lungs of 35 female Osborne-Mendel rats per group. To compare the carcinogenic potency of the heterocycles
On the metabolic activation of benz[a]acridine and benz[c]acridine by rat liver and lung microsomes.
J Jacob et al.
Cancer letters, 16(3), 297-306 (1982-09-01)
The metabolism of benz[a]- and benz[c]acridine by liver and lung microsomes from untreated, phenobarbital (PB)-treated and benzo[k]fluoranthene (BkF)-treated rats has been studied by gas chromatography/mass spectrometry (GC/MS). Epoxidation and hydrolysis of the epoxides to dihydrodiols were found to be the
J Molnár et al.
Anticancer research, 13(4), 1019-1025 (1993-07-01)
This review summarizes our experiments which are investigating the relationship between the structure and activity of mainly phenothiazines, benzo[a]phenothiazines and benz[c]acridines. Phenothiazines had potent antiplasmid and antibacterial activities, but induced weak antimicrobial activity in vivo. Their antiplasmid activity seemed to
N Motohashi et al.
Anticancer research, 12(4), 1207-1210 (1992-07-01)
The abilities of 14 phenothiazines, 8 benzo[a]phenothiazines and 12 benz[c]acridines to induce an antibacterial effect against Escherichia coli K12 were compared. Several phenothiazines, which showed antiplasmid activity, displayed the most potent antibacterial activity. All benz[c]acridine derivatives were moderately antibacterial, whereas
Benz[a]acridine.
IARC monographs on the evaluation of the carcinogenic risk of chemicals to humans, 32, 123-127 (1983-12-01)
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.
![Dibenz[a,h]acridine BCR®, certified reference material](/deepweb/assets/sigmaaldrich/product/structures/383/751/70b25872-405f-49b1-b76b-ed5e018ce265/640/70b25872-405f-49b1-b76b-ed5e018ce265.png)
![Benzo[h]quinoline 97%](/deepweb/assets/sigmaaldrich/product/structures/344/715/928932d2-4ca4-4402-b56c-85a80100ce17/640/928932d2-4ca4-4402-b56c-85a80100ce17.png)
![4H-benzo[def]carbazole AldrichCPR](/deepweb/assets/sigmaaldrich/product/structures/319/198/4208123a-456b-4f27-bc62-bc3af7c1d403/640/4208123a-456b-4f27-bc62-bc3af7c1d403.png)
![Dibenz[a,i]acridine BCR®, certified reference material](/deepweb/assets/sigmaaldrich/product/structures/531/045/5d4c722d-9928-44fb-8bbb-7f4e27442f63/640/5d4c722d-9928-44fb-8bbb-7f4e27442f63.png)
![11H-Benzo[a]carbazole](/deepweb/assets/sigmaaldrich/product/structures/391/065/abfb4cba-81ab-44b8-a816-d8791a903400/640/abfb4cba-81ab-44b8-a816-d8791a903400.png)
![Dibenz[c,h]acridine BCR®, certified reference material](/deepweb/assets/sigmaaldrich/product/structures/364/643/698df9fb-5b7d-467a-b47e-c8318e2ed298/640/698df9fb-5b7d-467a-b47e-c8318e2ed298.png)