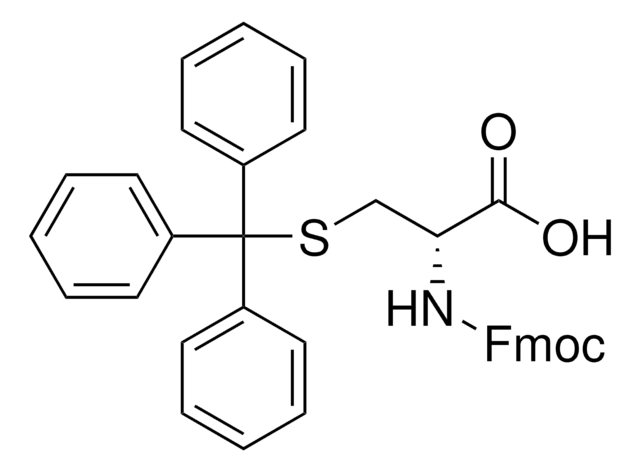
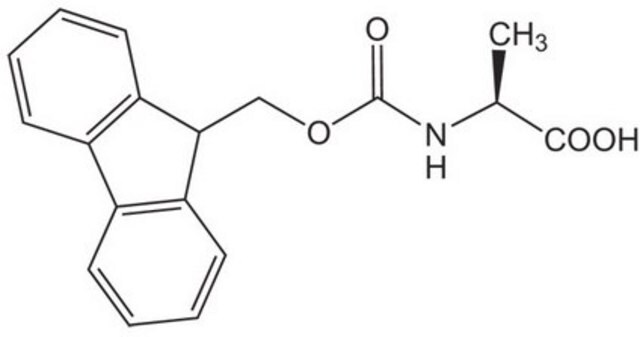
추천 제품
Quality Level
제품 라인
Novabiochem®
분석
≥98% (TLC)
≥98.0% (acidimetric)
≥99.0% (HPLC)
양식
powder
반응 적합성
reaction type: Fmoc solid-phase peptide synthesis
제조업체/상표
Novabiochem®
mp
164-175 °C
응용 분야
peptide synthesis
작용기
thiol
저장 온도
−20°C (−15°C to −25°C)
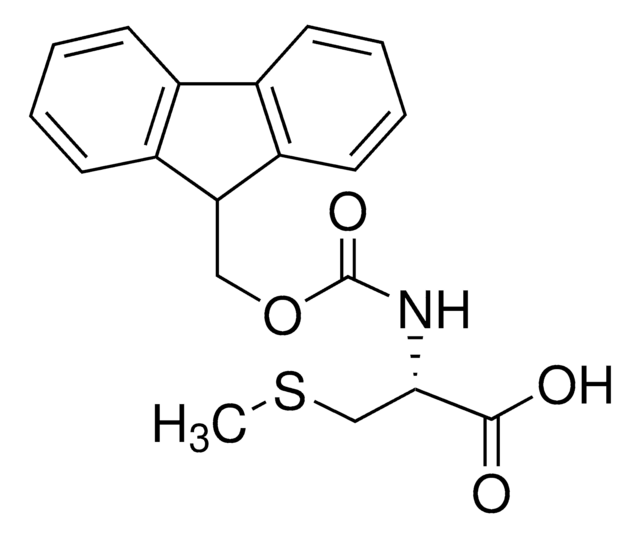
SMILES string
S(C[C@H](NC(=O)OCC4c5c(cccc5)c6c4cccc6)C(=O)[O-])C(c3ccccc3)(c2ccccc2)c1ccccc1
InChI
1S/C37H31NO4S/c39-35(40)34(38-36(41)42-24-33-31-22-12-10-20-29(31)30-21-11-13-23-32(30)33)25-43-37(26-14-4-1-5-15-26,27-16-6-2-7-17-27)28-18-8-3-9-19-28/h1-23,33-34H,24-25H2,(H,38,41)(H,39,40)/p-1/t34-/m0/s1
InChI key
KLBPUVPNPAJWHZ-UMSFTDKQSA-M
일반 설명
High purity Fmoc-protected amino acid for research and process production of peptides, with very low levels of dipeptide, free-amino acids and acetic acid impurities. The standard derivative for Fmoc SPPS of peptides containing Cys [1]. The Trt group is removed with 95% TFA containing 1-5% TIS. Ideally, this derivative should be introduced using the symmetrical anhydride or DIPCDI/HOBt activation [2,3] to minimize enantiomerization. If activation with uronium or phosphonium reagents, such as HBTU or PyBOP®, is to be employed, it is strongly recommended that collidine is used as the base [4], as this has been shown to significantly reduce loss of optical integrity during coupling.
Associated Protocols and Technical Articles
Fmoc-amino acids for Peptide Production
Cleavage and Deprotection Protocols for Fmoc SPPS
Fmoc SPPS of Cysteine-Containing Peptides
Literature references
[1] S. N. McCurdy (1989) Pept. Res., 2, 147.
[2] T. Kaiser, et al. (1996) Tetrahedron Lett., 37, 1187.
[3] Y. X. Han, et al. (1997) J. Org. Chem., 62, 4307.
[4] Y. N. Angell (2002) J. Peptide Res., 5, 292.
Associated Protocols and Technical Articles
Fmoc-amino acids for Peptide Production
Cleavage and Deprotection Protocols for Fmoc SPPS
Fmoc SPPS of Cysteine-Containing Peptides
Literature references
[1] S. N. McCurdy (1989) Pept. Res., 2, 147.
[2] T. Kaiser, et al. (1996) Tetrahedron Lett., 37, 1187.
[3] Y. X. Han, et al. (1997) J. Org. Chem., 62, 4307.
[4] Y. N. Angell (2002) J. Peptide Res., 5, 292.
애플리케이션
- On-resin synthesis of cyclic peptides via tandem N-to-S acyl migration and intramolecular thiol additive-free native chemical ligation: Discusses the use of Fmoc-Cys(Trt)-OH in the synthesis of cyclic peptides, highlighting the efficiency of the resin synthesis method. (Serra et al., 2020).
- Selective Bi‐directional Amide Bond Cleavage of N‐Methylcysteinyl Peptide: The study utilized Fmoc-Cys(Trt)-OH in exploring selective bi-directional amide bond cleavage in peptides, providing insights into controlled peptide modification. (Qiu et al., 2014).
결합
Replaces: 04-12-1018
분석 메모
Colour (visual): white to off white
Appearance of substance (visual): powder
Colour index (0,5 M in DMF): ≤ 150 Hazen
Identity (IR): passes test
Enantiomeric purity: ≥ 99.5 % (a/a)
Purity (HPLC): ≥ 99.0 % (a/a)
Fmoc-ß-Ala-OH (HPLC): ≤ 0.1 % (a/a)
Fmoc-ß-Ala-Cys (Trt) -OH (HPLC): ≤ 0.1 % (a/a)
Fmoc-Cys(Trt)-Cys(Trt)-OH (HPLC): ≤ 0.1 % (a/a)
Fmoc-Cys-OH (HPLC): ≤ 0.1 % (a/a)
Assay free amino acid (HPLC): ≤ 0.2 %
Purity (TLC(011A)): ≥ 98 %
Purity (TLC(0811)): ≥ 98 %
Solubility (1 mmole in 2 ml DMF): clearly soluble
Assay (acidimetric): ≥ 98.0 %
Water (K. F.): ≤ 2.0 %
Ethyl acetate (HS-GC): ≤ 0.5 %
Acetate (IC): ≤ 0.02 %
To see the solvent systems used for TLC of Novabiochem® products please click here.
Appearance of substance (visual): powder
Colour index (0,5 M in DMF): ≤ 150 Hazen
Identity (IR): passes test
Enantiomeric purity: ≥ 99.5 % (a/a)
Purity (HPLC): ≥ 99.0 % (a/a)
Fmoc-ß-Ala-OH (HPLC): ≤ 0.1 % (a/a)
Fmoc-ß-Ala-Cys (Trt) -OH (HPLC): ≤ 0.1 % (a/a)
Fmoc-Cys(Trt)-Cys(Trt)-OH (HPLC): ≤ 0.1 % (a/a)
Fmoc-Cys-OH (HPLC): ≤ 0.1 % (a/a)
Assay free amino acid (HPLC): ≤ 0.2 %
Purity (TLC(011A)): ≥ 98 %
Purity (TLC(0811)): ≥ 98 %
Solubility (1 mmole in 2 ml DMF): clearly soluble
Assay (acidimetric): ≥ 98.0 %
Water (K. F.): ≤ 2.0 %
Ethyl acetate (HS-GC): ≤ 0.5 %
Acetate (IC): ≤ 0.02 %
To see the solvent systems used for TLC of Novabiochem® products please click here.
법적 정보
Novabiochem is a registered trademark of Merck KGaA, Darmstadt, Germany
PyBOP is a registered trademark of Merck KGaA, Darmstadt, Germany
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
시험 성적서(COA)
제품의 로트/배치 번호를 입력하여 시험 성적서(COA)을 검색하십시오. 로트 및 배치 번호는 제품 라벨에 있는 ‘로트’ 또는 ‘배치’라는 용어 뒤에서 찾을 수 있습니다.
이미 열람한 고객
관련 콘텐츠
Purer Fmocs Means Purer Peptides
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.