1.00894
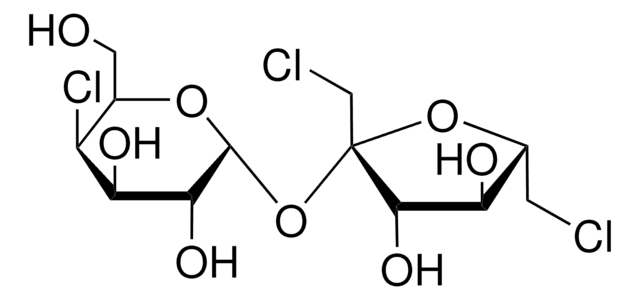
Sucralose
powder, EMPROVE® ESSENTIAL, Ph. Eur., ChP, JPE, NF
의약품 제조
동의어(들):
Sucralose, 1,6-Dichloro-1,6-dideoxy-β-D-fructofuranosyl-4-chloro-4-deoxy-α-D-galactopyranoside, E955, Trichlorosucrose
About This Item
추천 제품
Agency
ChP
JPE
NF
Ph. Eur.
Quality Level
설명
EMPROVE® ESSENTIAL Ph Eur,NF,JPE
제품 라인
EMPROVE® ESSENTIAL
양식
fine powder
powder
입자 크기
≤12 μm (d90)
pH
6-8 (20 °C, 100 g/L in H2O)
mp
130 °C (decomposition)
density
1.62 g/cm3 at 20 °C
응용 분야
liquid formulation
pharmaceutical
solid formulation
저장 온도
15-25°C
SMILES string
OC[C@H]1O[C@H](O[C@]2(CCl)O[C@H](CCl)[C@@H](O)[C@@H]2O)[C@H](O)[C@@H](O)[C@H]1Cl
InChI
1S/C12H19Cl3O8/c13-1-4-7(17)10(20)12(3-14,22-4)23-11-9(19)8(18)6(15)5(2-16)21-11/h4-11,16-20H,1-3H2/t4-,5-,6+,7-,8+,9-,10+,11-,12+/m1/s1
InChI key
BAQAVOSOZGMPRM-QBMZZYIRSA-N
유사한 제품을 찾으십니까? 방문 제품 비교 안내
일반 설명
As part of our EMPROVE® Program, our raw materials are offered with extensive documentation facilitating compliance of your pharma and biopharma product, full supply chain transparency and risk mitigation. Our SAFC® portfolio of high-quality products for biopharmaceutical and pharmaceutical formulation and production withstands strict quality control procedures and is produced according to applicable cGMP guidelines.
애플리케이션
Our Sucralose is available in micronized or granular form, both pharmaceutical grade, allowing you to choose depending on your individual needs. Due to its optimal dispersion behavior, sucralose powder is typically recommended for solid dose formulations, while the fast dissolution behavior of sucralose granular makes it very well-suited for liquid formulations.
생화학적/생리학적 작용
특징 및 장점
- Excellent sugar-like taste profiles
- Masking bitter aftertaste and medical off-notes with ease
- High sweetness potency offers improved cost-efficiency
- Enhanced stability
- Non-cariogenic and suitable for diabetics and patients with phenylketonuria
법적 정보
면책조항
Storage Class Code
11 - Combustible Solids
WGK
WGK 2
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
시험 성적서(COA)
제품의 로트/배치 번호를 입력하여 시험 성적서(COA)을 검색하십시오. 로트 및 배치 번호는 제품 라벨에 있는 ‘로트’ 또는 ‘배치’라는 용어 뒤에서 찾을 수 있습니다.
이미 열람한 고객
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.