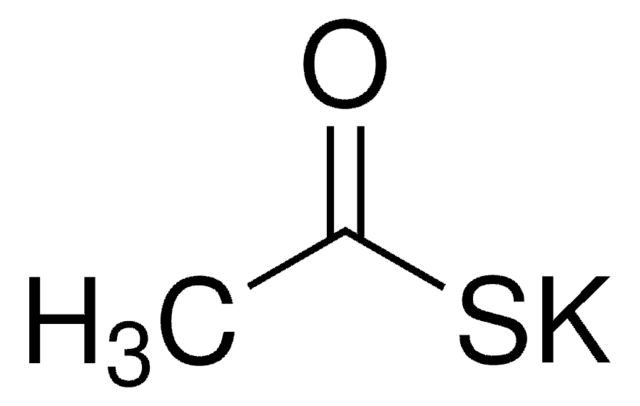
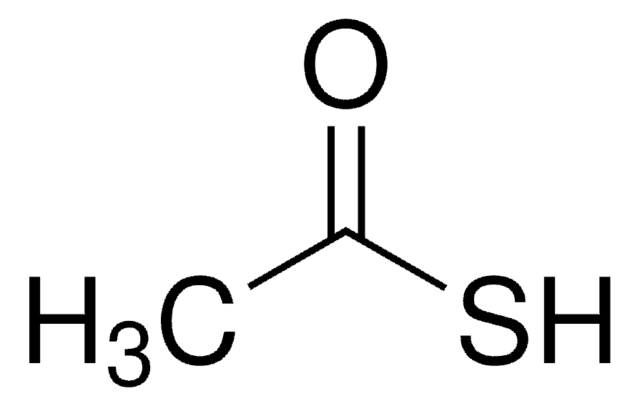
추천 제품
애플리케이션
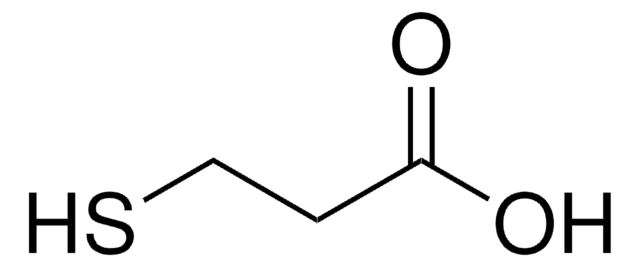
Thioacetic acid (TAA) can undergo:
- Enantioselective addition to nitroalkenes to form chiral 1,2-aminothiol derivatives in the presence of a novel sulfinyl urea organocatalyst. This method has been successfully employed in the synthesis of antifungal drug, sulconazole.
- Asymmetric Michael addition reaction with chalcones in the presence of a bifunctional amine thiourea catalyst to form synthetically useful thioesters.
- Asymmetric 1,6-conjugate addition with para-quinone methides in the presence of a chiral phosphoric acid catalyst to form chiral sulfur-containing diphenylmethane-type compounds.
- Conjugate addition to methacrylamides with chiral trans-2,5-disubstituted pyrrolidine auxiliaries to form chiral β-mercaptocarboxylic acid derivatives.
Thioacetic acid is a reagent for introduction of the thiol group into organic molecules.
신호어
Danger
유해 및 위험 성명서
Hazard Classifications
Acute Tox. 3 Oral - Acute Tox. 4 Inhalation - Eye Dam. 1 - Flam. Liq. 2 - Skin Sens. 1
Storage Class Code
3 - Flammable liquids
WGK
WGK 3
Flash Point (°F)
64.4 °F
Flash Point (°C)
18 °C
개인 보호 장비
Faceshields, Gloves, Goggles, type ABEK (EN14387) respirator filter
시험 성적서(COA)
제품의 로트/배치 번호를 입력하여 시험 성적서(COA)을 검색하십시오. 로트 및 배치 번호는 제품 라벨에 있는 ‘로트’ 또는 ‘배치’라는 용어 뒤에서 찾을 수 있습니다.
이미 열람한 고객
Phosphoric Acid Catalyzed Asymmetric 1, 6?Conjugate Addition of Thioacetic Acid to para?Quinone Methides.
Dong N, et al.
Angewandte Chemie (International Edition in English), 55(4), 1460-1464 (2016)
Organocatalytic enantioselective Michael addition of thioacetic acid to enones.
Li Hao, et al.
Tetrahedron Letters, 47(18), 3145-3148 (2006)
Asymmetric induction in the conjugate addition of thioacetic acid to methacrylamides with chiral auxiliaries.
Kim B H, et al.
Tetrahedron Asymmetry, 16(6), 1215-1220 (2005)
Christina Wedemeyer-Exl et al.
Organic & biomolecular chemistry, 5(13), 2119-2128 (2007-06-22)
The thiol-dependent methylation of heptamethyl cob(II)yrinate 8r with methyl iodide and methyl tosylate was explored under a variety of conditions. The interaction of the heptamethyl cob(II)yrinate with a variety of thiols was monitored prior to the addition of the methylating
Jianhua Ren
The journal of physical chemistry. A, 110(50), 13405-13411 (2006-12-15)
We studied the effects of polar groups on the gas-phase acidities of carboxylic acids experimentally and computationally. In this connection, the gas-phase acidities (DeltaH(acid), the enthalpy of deprotonation, and DeltaG(acid), the deprotonation free energy) of borane-complexed methylaminoacetic acid ((CH(3))2N(BH(3))CH(2)CO(2)H) and
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.