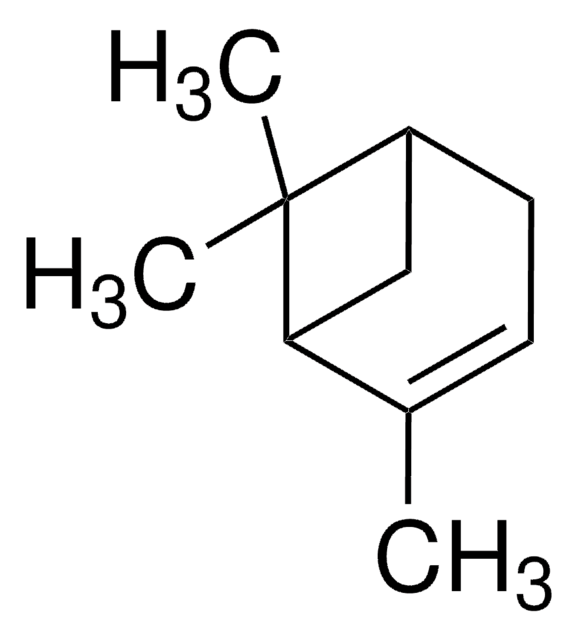
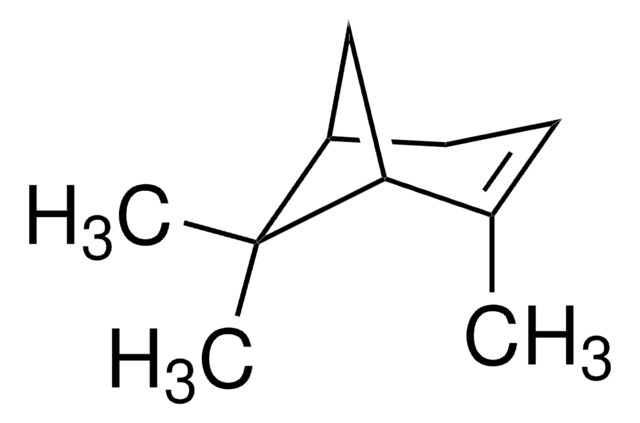
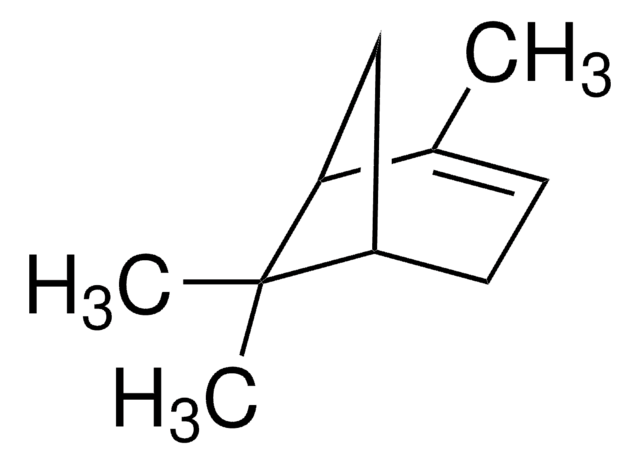
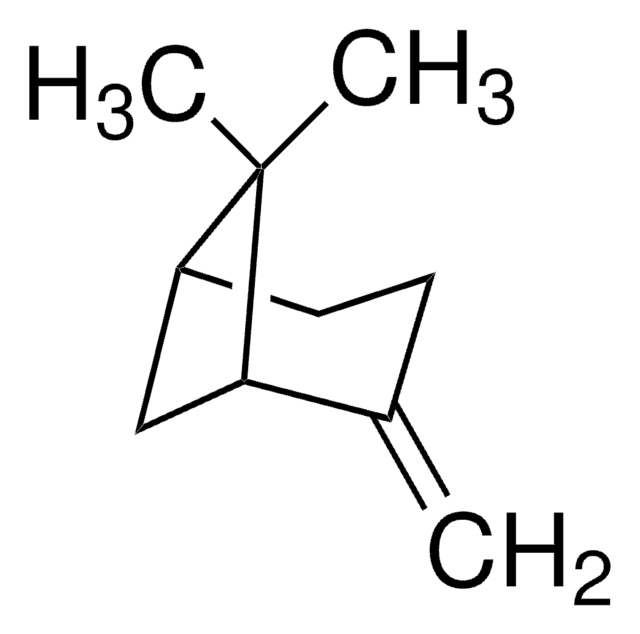
추천 제품
Quality Level
분석
98%
광학 활성
[α]/D 35 to 54, neat
광학 순도
ee: ≥85% (GLC)
autoignition temp.
491 °F
refractive index
n20/D 1.465 (lit.)
bp
155-156 °C (lit.)
mp
−62 °C (lit.)
density
0.858 g/mL at 20 °C (lit.)
SMILES string
CC1=CC[C@@H]2C[C@H]1C2(C)C
InChI
1S/C10H16/c1-7-4-5-8-6-9(7)10(8,2)3/h4,8-9H,5-6H2,1-3H3/t8-,9-/m1/s1
InChI key
GRWFGVWFFZKLTI-RKDXNWHRSA-N
유사한 제품을 찾으십니까? 방문 제품 비교 안내
일반 설명
(+)-α-Pinene, a monoterpene, is a bio-jet fuel precursor. It undergoes ozonolysis to produce secondary organic aerosol (SOA) mass.
애플리케이션
(+)-α-Pinene can be used:
- To prepare chiral organoboranes, which are used in the synthesis of optically active compounds and in Suzuki coupling.
- In the synthesis of chiral pyridine-type N-oxides as catalysts for the allylation of aromatic aldehydes.
- To synthesize 2,3-pinane diamine, a chiral auxiliary in several catalysts and reagents.
(+)-α-Pinene has been used in a tolerance testing procedure to detect the genes in Marinobacter aquaeolei, for engineering a pinene tolerant Escherichia coli.
신호어
Danger
유해 및 위험 성명서
Hazard Classifications
Aquatic Acute 1 - Aquatic Chronic 1 - Asp. Tox. 1 - Flam. Liq. 3 - Skin Irrit. 2 - Skin Sens. 1
Storage Class Code
3 - Flammable liquids
WGK
WGK 3
Flash Point (°F)
91.4 °F - closed cup
Flash Point (°C)
33 °C - closed cup
개인 보호 장비
Eyeshields, Faceshields, Gloves, type ABEK (EN14387) respirator filter
이미 열람한 고객
Coating of soot and (NH4)2SO4 particles by ozonolysis products of a-pinene.
Saathoff, H., et al.
Journal of Aerosol Science, 34(10), 1297-1321 (2003)
Synthesis and characterization of novel chiral 1, 2-diamines derived from α-pinene.
Suzuki T, et al.
Chemistry Letters (Jpn), 34(11), 1476-1477 (2005)
Engineering improved bio-jet fuel tolerance in Escherichia coli using a transgenic library from the hydrocarbon-degrader Marinobacter aquaeolei.
Tomko TA and Dunlop MJ.
Biotechnology for Biofuels, 8(1), 165-165 (2015)
Multistep synthesis of complex boronic acids from simple MIDA boronates.
Gillis E P and Burke M D
Journal of the American Chemical Society, 130(43), 14084-14085 (2008)
Chiral synthesis via organoboranes. 18. Selective reductions. 43. Diisopinocampheylchloroborane as an excellent chiral reducing reagent for the synthesis of halo alcohols of high enantiomeric purity. A highly enantioselective synthesis of both optical isomers of Tomoxetine, Fluoxetine, and Nisoxetine.
Srebnik M, et al.
The Journal of Organic Chemistry, 53(13), 2916-2920 (1988)
Chromatograms
suitable for GCsuitable for GCsuitable for GC자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.