추천 제품
분석
98%
형태
powder
mp
>300 °C (lit.)
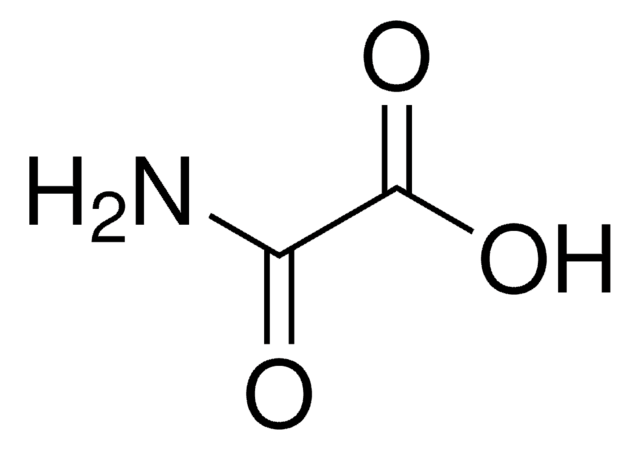
SMILES string
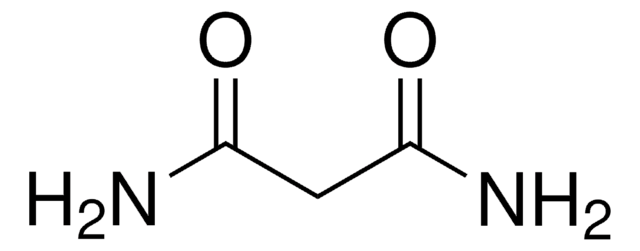
NC(=O)C(N)=O
InChI
1S/C2H4N2O2/c3-1(5)2(4)6/h(H2,3,5)(H2,4,6)
InChI key
YIKSCQDJHCMVMK-UHFFFAOYSA-N
유사한 제품을 찾으십니까? 방문 제품 비교 안내
애플리케이션
Oxamide can be used:
- As a precursor for the synthesis of ligands such as bis(benzimidazole) and Schiff base ligands formed by condensation with furfural.
- Carbon nitride (g-C3N4) nanotubes by self-assembly polymerization with urea.
- As a bridging ligand for the synthesis of binuclear IrIII complex [Ir2(μ2-oxamidato-N,N′,O,O′)(ptpy)4], ptpy = 2-(p-tolyl)pyridinato.
신호어
Warning
유해 및 위험 성명서
Hazard Classifications
Acute Tox. 4 Oral - Eye Irrit. 2 - Skin Irrit. 2
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
개인 보호 장비
dust mask type N95 (US), Eyeshields, Gloves
이미 열람한 고객
T L Nguyen et al.
Journal of the American Chemical Society, 123(44), 11057-11064 (2001-11-01)
Ureas characteristically form one-dimensional hydrogen-bonded alpha-networks with a repeat distance of about 4.60 A. Oxamides form similar alpha-networks with a longer 5.05 A repeat distance. The urea of glycine and the oxamide of glycine were each cocrystallized with a series
Advancing the n?π* electron transition of carbon nitride nanotubes for H2 photosynthesis
Zhang G, et al.
Journal of Material Chemistry A, 5(25), 12723-12728 (2017)
Bis (benzimidazole) as supramolecular building block in manganese (IV) chemistry
Samol'ova E, et al.
Journal of Molecular Structure, 1176, 366-375 (2019)
E Armelin et al.
The Journal of organic chemistry, 66(24), 8076-8085 (2001-11-28)
The conformational properties of the oxalamide group and crystal structure of several polyoxalamides have been investigated by computational methods. First, a detailed quantum mechanical study of the conformational preferences of N,N'-dimethyloxalamide is reported. Results, which were obtained at the MP2/6-31G(d)
Synthesis, characterization, and biological and anticancer studies of mixed ligand complexes with Schiff base and 2, 2?-bipyridine
Omar MM, et al.
Applied Organometallic Chemistry, 31(10), e3724-e3724 (2017)
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.