추천 제품
Quality Level
분석
98%
양식
liquid
refractive index
n20/D 1.595 (lit.)
bp
230 °C (lit.)
mp
16-19 °C (lit.)
density
1.585 g/mL at 25 °C (lit.)
SMILES string
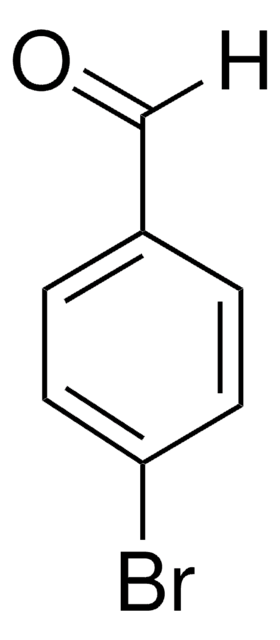
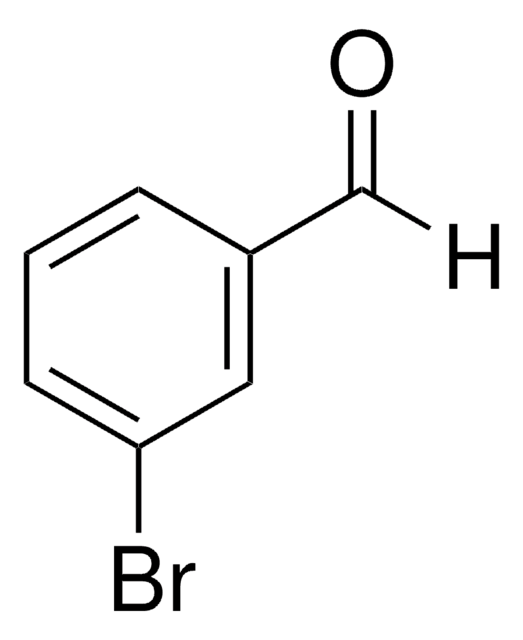
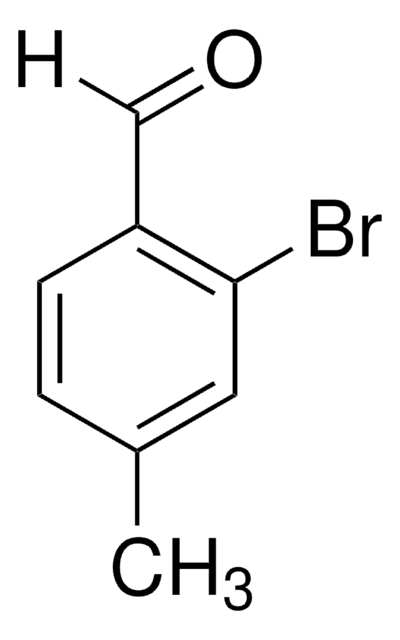
Brc1ccccc1C=O
InChI
1S/C7H5BrO/c8-7-4-2-1-3-6(7)5-9/h1-5H
InChI key
NDOPHXWIAZIXPR-UHFFFAOYSA-N
유사한 제품을 찾으십니까? 방문 제품 비교 안내
일반 설명
2-Bromobenzaldehyde is a versatile building block used in Suzuki-Miyaura and Buchwald-Hartwig reactions, cross-coupling reactions to form carbon-carbon or carbon-heteroatom bonds
애플리케이션
Synthetic applications of 2-bromobenzaldehyde include:
- Synthesis of aza-fused polycyclic quinolines through copper-catalyzed cascade reaction.
- Preparation of 1-substituted indazoles by CuI-catalyzed coupling with N-aryl hydrazides.
- It is a key starting material in the total synthesis of an anticancer agent, (-)-taxol.
- Palladium/copper-catalyzed coupling and cyclization of terminal acetylenes derived from 2-bromobenzaldehyde and unsaturated imines can be used in the synthesis of wide range of isoquinolines, including decumbenine B.
- Fluoren-9-ones can be synthesized by annulation of arynes, generated in situ from 2-(trimethylsilyl)aryl triflates, with 2-bromobenzaldehyde in the presence of palladium(0) catalyst.
- It can also be used to build a variety of steroid frameworks, in which ring A is derived from 2-bromobenzaldehyde.
신호어
Warning
유해 및 위험 성명서
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
표적 기관
Respiratory system
Storage Class Code
10 - Combustible liquids
WGK
WGK 3
Flash Point (°F)
203.0 °F - closed cup
Flash Point (°C)
95 °C - closed cup
개인 보호 장비
Eyeshields, Gloves, type ABEK (EN14387) respirator filter
가장 최신 버전 중 하나를 선택하세요:
시험 성적서(COA)
Lot/Batch Number
이미 열람한 고객
Synthesis of isoquinolines and pyridines by the palladium/copper-catalyzed coupling and cyclization of terminal acetylenes and unsaturated imines: the total synthesis of decumbenine B.
Roesch K R and Larock R C
The Journal of Organic Chemistry, 67(1), 86-94 (2002)
Enantioselective total synthesis of (?)-taxol.
Kusama H, et al.
Journal of the American Chemical Society, 122(16), 3811-3820 (2000)
Assembly of N, N-Disubstituted Hydrazines and 1-Aryl-1 H-indazoles via Copper-Catalyzed Coupling Reactions.
Xiong X, et al.
Organic Letters, 14(10), 2552-2555 (2012)
Synthesis of aza-fused polycyclic quinolines through copper-catalyzed cascade reactions.
Cai Q, et al.
Organic Letters, 12(7), 1500-1503 (2010)
A gold-catalyzed domino process to the steroid framework.
Hildebrandt D and Dyker G
The Journal of Organic Chemistry, 71(18), 6728-6733 (2006)
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.