모든 사진(2)
About This Item
실험식(Hill 표기법):
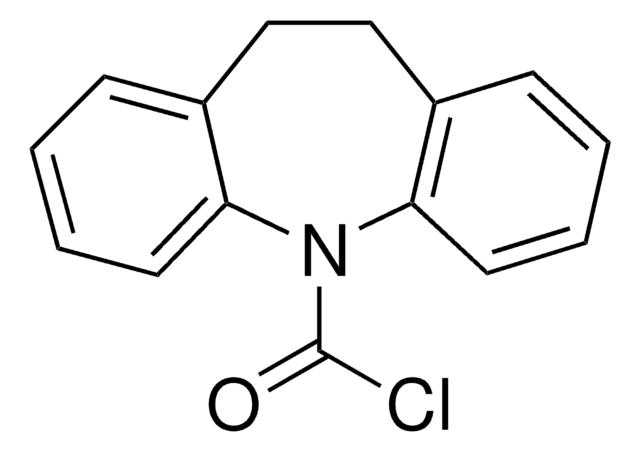
C15H10ClNO
CAS Number:
Molecular Weight:
255.70
MDL number:
UNSPSC 코드:
12352100
PubChem Substance ID:
NACRES:
NA.22
추천 제품
Quality Level
분석
90%
mp
149-153 °C (lit.)
작용기
chloro
SMILES string
ClC(=O)N1c2ccccc2C=Cc3ccccc13
InChI
1S/C15H10ClNO/c16-15(18)17-13-7-3-1-5-11(13)9-10-12-6-2-4-8-14(12)17/h1-10H
InChI key
APJYHXJGXDPGBA-UHFFFAOYSA-N
유사한 제품을 찾으십니까? 방문 제품 비교 안내
일반 설명
Dibenz [b,f]azepine-5-carbonyl chloride or 5H-dibenz [b,f]azepine-5-carbonyl chloride is a tricyclic heterocyclic compound that can be synthesized from 5H-dibenz[ b,f]azepine.
애플리케이션
Dibenz [b,f]azepine-5-carbonyl chloride may be used in the preparation of trans-10,11-dibromo-10,11-dihydro-5H-dibenz[b,f]azepine-5-carbonyl chloride via bromination using bromine. It may also be used to prepare urea derivatives, which are potent P2X4 receptor (purinergic receptor) antagonists.
신호어
Danger
유해 및 위험 성명서
Hazard Classifications
Skin Corr. 1B
Storage Class Code
8A - Combustible corrosive hazardous materials
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
개인 보호 장비
Eyeshields, Faceshields, Gloves, type P3 (EN 143) respirator cartridges
Maoqun Tian et al.
Bioorganic & medicinal chemistry, 22(3), 1077-1088 (2014-01-15)
Antagonists for the P2 receptor subtype P2X4, an ATP-activated cation channel receptor, have potential as novel drugs for the treatment of neuropathic pain and other inflammatory diseases. In the present study, a series of 47 carbamazepine derivatives including 32 novel
Kinetic evidence for rate determination during the nucleophilic step of olefin bromination. The case of 5H-dibenz [b, f] azepine-5-carbonyl chloride.
Bellucci G and Chiappe C.
The Journal of Organic Chemistry, 58(25), 7120-7127 (1993)
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.
![10,11-Dihydro-5H-dibenz[b,f]azepine 97%](/deepweb/assets/sigmaaldrich/product/structures/282/468/27ed6f23-3d01-4628-8293-f0051a6f3b7c/640/27ed6f23-3d01-4628-8293-f0051a6f3b7c.png)