추천 제품
분석
97%
양식
powder
반응 적합성
core: iridium
reagent type: catalyst
mp
160-165 °C (dec.) (lit.)
SMILES string
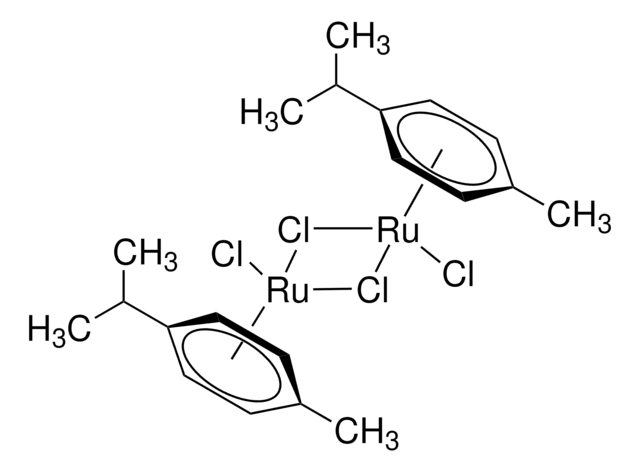
Cl[Ir].Cl[Ir].[CH]1[CH]CCCCCC1.[CH]2[CH]CCCCCC2.[CH]3[CH]CCCCCC3.[CH]4[CH]CCCCCC4
InChI
1S/4C8H14.2ClH.2Ir/c4*1-2-4-6-8-7-5-3-1;;;;/h4*1-2H,3-8H2;2*1H;;/q;;;;;;2*+1/p-2/b4*2-1-;;;;
InChI key
WBRREXQCZAFSKS-XFCUKONHSA-L
애플리케이션
Catalyst for:
- Isomerization-hydroboration reactions with nido-carboranyldiphosphine as stabilizing ligand
- Hydrogen peroxide oxidation of hydroxamic acids and their subsequent hetero Diels-Alder cycloaddition reactions
- Immobilization of organic functional groups onto solid supports through vinylsilane coupling reactions
- Alkylation reactions
- Guerbet reaction
- Allylic amination reactions in a DNA-diene-iridium(I) hybrid system
- Asymmetric hydroamination reactions
Chlorobis(cyclooctene)iridium(I)dimer can be used as a catalyst for:
- Isomerization-hydroboration reactions with nido-carboranyldiphosphine as stabilizing ligand.
- Hydrogen peroxide oxidation of hydroxamic acids and their subsequent hetero Diels-Alder cycloaddition reactions.
- Immobilization of organic functional groups onto solid supports through vinylsilane coupling reactions.
- Alkylation reactions.
- Guerbet reaction.
- Allylic amination reactions using a DNA-diene-iridium(I) hybrid system.
- Asymmetric hydroamination reactions.
신호어
Warning
유해 및 위험 성명서
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
표적 기관
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
개인 보호 장비
dust mask type N95 (US), Eyeshields, Gloves
이미 열람한 고객
An efficient and recyclable catalytic system comprising nano-iridium (0) and a pyridinium salt of nido-carboranyldiphosphine for the synthesis of one-dimensional boronate esters via hydroboration reaction.
Zhu Y, et al.
Organometallics, 31(7), 2589-2596 (2011)
Alkylation of active methylene compounds with alcohols catalyzed by an iridium complex.
Morita M, et al.
Chemical Communications (Cambridge, England), 27, 2850-2852 (2007)
Allylic amination by a DNA?diene?iridium (I) hybrid catalyst.
Fournier P, et al.
Angewandte Chemie (International Edition in English), 48(24), 4426-4429 (2009)
Transition-metal-catalyzed immobilization of organic functional groups onto solid supports through vinylsilane coupling reactions.
Park J W and Jun C H
Journal of the American Chemical Society, 132(21), 7268-7269 (2010)
Iridium (I)-catalyzed hydrogen peroxide oxidation of hydroxamic acids and hetero Diels?Alder reaction of the acyl nitroso intermediates with cyclopentadiene.
Iwasa S, et al.
Tetrahedron Letters, 43(35), 6159-6161 (2002)
문서
Arylboronic acids and esters are invaluable tools for the chemical community. These powerful reagents are used for a variety of transformations, most notably the Suzuki-Miyaura cross-coupling reaction.
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.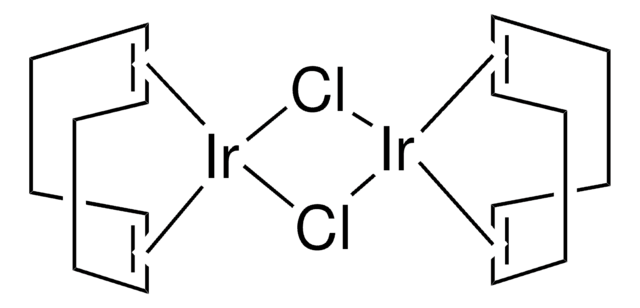




![[Ir(cod)(acac)] Umicore](/deepweb/assets/sigmaaldrich/product/structures/188/615/470bfca9-6b61-476a-9486-f7da61962e4c/640/470bfca9-6b61-476a-9486-f7da61962e4c.png)