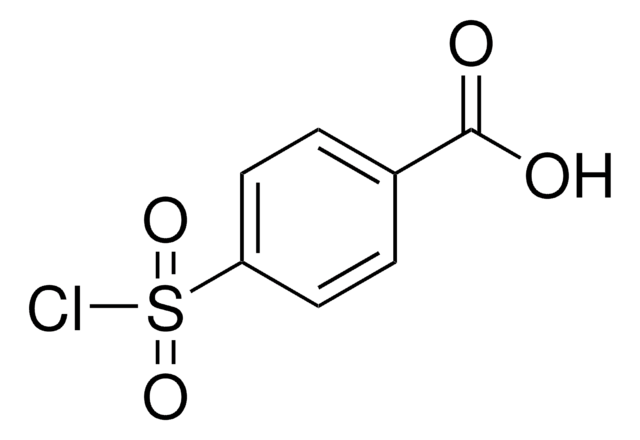
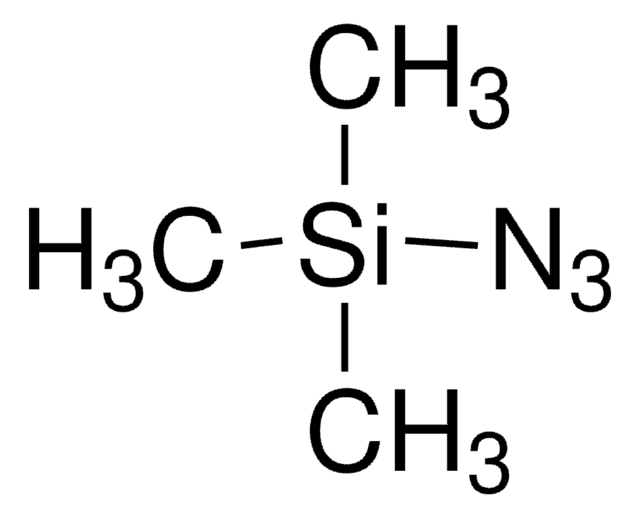About This Item
추천 제품
Quality Level
분석
97%
반응 적합성
reaction type: click chemistry
mp
180 °C (dec.) (lit.)
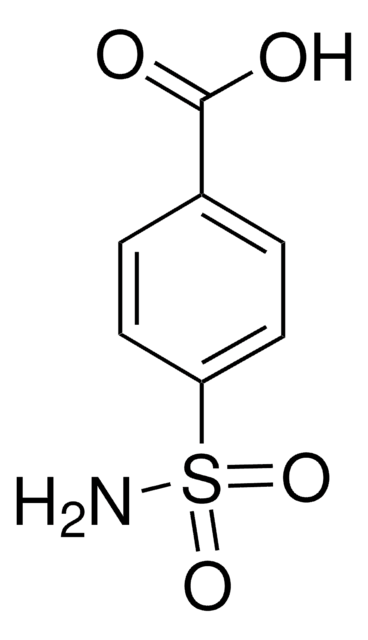
작용기
azide
carboxylic acid
저장 온도
2-8°C
SMILES string
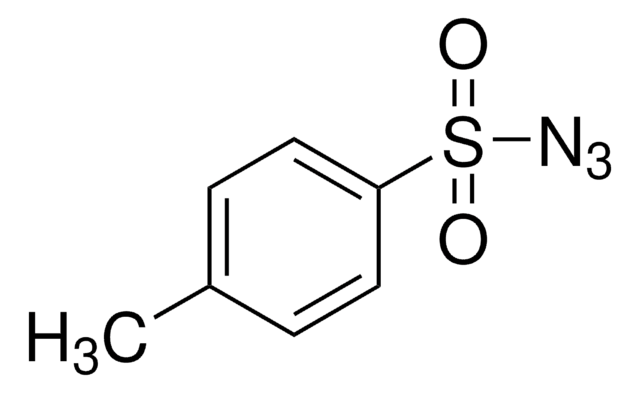
OC(=O)c1ccc(cc1)S(=O)(=O)N=[N+]=[N-]
InChI
1S/C7H5N3O4S/c8-9-10-15(13,14)6-3-1-5(2-4-6)7(11)12/h1-4H,(H,11,12)
InChI key
OWULJVXJAZBQLL-UHFFFAOYSA-N
애플리케이션
Synthesis of anti-inflammatory agents
Azide amidation
Reactions of thio acids with azides
Chemoselective sodium borohydride reduction of azides in water
Reagent for:
Photo-Stevens rearrangement
Cobalt-catalyzed synthesis of tertiary azides
신호어
Warning
유해 및 위험 성명서
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
표적 기관
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
개인 보호 장비
dust mask type N95 (US), Eyeshields, Gloves
문서
The chemistry of organoazides is exceedingly rich, since the azide functionality reacts with electrophiles, nucleophiles, and dipolarophiles, with or without the extrusion of dinitrogen. Common place transformation such as Staudinger reductions or ligations, Cu(I)-catalyzed Huisgen cycloadditions (of the “click” reaction family), Curtius or Schmidt rearrangents, nitrene reactions, or imine formation via aza-Wittig reactions all necessitate organoazide precursors or intermediates
Since the preparation of the first organic azide, phenyl azide, by Peter Griess in 1864 this energy-rich and versatile class of compounds has enjoyed considerable interest.
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.