303232
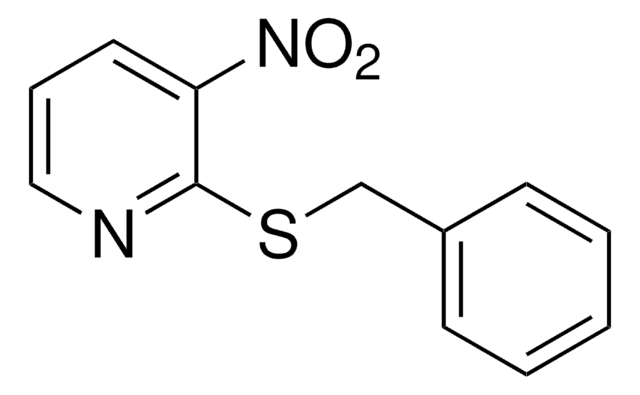
3-Nitro-2-pyridinesulfenyl chloride
95%
동의어(들):
(3-Nitro-2-pyridyl)sulfenyl chloride, 2-(Chlorosulfanyl)-3-nitropyridine, 3-Nitropyridinyl-2-sulfenyl chloride, [(3-Nitropyridin-2-yl)sulfanyl]chlorane
로그인조직 및 계약 가격 보기
모든 사진(2)
About This Item
실험식(Hill 표기법):
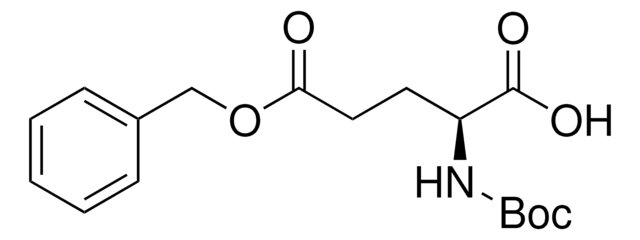
C5H3ClN2O2S
CAS Number:
Molecular Weight:
190.61
MDL number:
UNSPSC 코드:
12352100
PubChem Substance ID:
NACRES:
NA.22
추천 제품
Quality Level
분석
95%
mp
205 °C (dec.) (lit.)
solubility
dichloromethane: soluble(lit.)
작용기
nitro
저장 온도
2-8°C
SMILES string
[O-][N+](=O)c1cccnc1SCl
InChI
1S/C5H3ClN2O2S/c6-11-5-4(8(9)10)2-1-3-7-5/h1-3H
InChI key
WTKQMHWYSBWUBE-UHFFFAOYSA-N
일반 설명
The 3-nitro-2-pyridinesulphenyl (Npys) moiety is useful as a protecting-activating group for cysteine, particularly in the synthesis of cyclic and unsymmetrical disulfides. The stability of NpysCl was studied in various solvents.
애플리케이션
3-Nitro-2-pyridinesulfenyl chloride (NpysCl) was employed as the starting material for the synthesis of N-, O- and S-Npys-protected amino acid.
신호어
Danger
유해 및 위험 성명서
Hazard Classifications
Eye Dam. 1 - Skin Corr. 1B
Storage Class Code
8A - Combustible corrosive hazardous materials
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
개인 보호 장비
Eyeshields, Faceshields, Gloves, type P3 (EN 143) respirator cartridges
가장 최신 버전 중 하나를 선택하세요:
K C Pugh et al.
International journal of peptide and protein research, 42(2), 159-164 (1993-08-01)
3-Nitro-2-pyridinesulfenyl chloride (NpysCl) is the starting material for the synthesis of N-, O- and S-Npys-protected amino acids. Two efficient, novel synthetic routes to NpysCl are described. The stability of NpysCl was determined in a variety of solvents, with and without
Y Shimohigashi et al.
Journal of chromatography, 597(1-2), 425-428 (1992-04-24)
The thiol groups of leucinthiol, cysteamine and cysteine incorporated into opioid peptides enkephalin and morphiceptin were activated by the 3-nitro-2-pyridinesulphenyl (Npys) group to form mixed disulphides highly reactive to a free thiol. Enkephalin analogues containing Npys-leucinthiol or -cysteine at positions
Alisson L Matsuo et al.
Biochemical and biophysical research communications, 355(4), 1000-1005 (2007-03-03)
The inhibitory capacity of C-Npys (S-[3-nitro-2-pyridinesulfenyl]) derivatives over thiol-containing serine proteases has never been tested. In the present work we used an extracellular serine-thiol proteinase activity from the fungal pathogen Paracoccidioides brasiliensis (PbST) to describe a potent inhibitory capacity of
R Matsueda et al.
Peptide research, 5(5), 262-264 (1992-09-01)
Two recent reports on the partial lability of the 3-nitro-2-pyridinesulfenyl (Npys) thiol protecting group towards 1-hydroxy-benzotriazole (HOBt) have prompted a rechecking of the chemical behavior of this group. Using both soluble and polymer-bound forms of Cys(Npys) as test materials, the
R G Simmonds et al.
International journal of peptide and protein research, 43(4), 363-366 (1994-04-01)
The 3-nitro-2-pyridinesulphenyl (Npys) moiety is finding increasing utility as a protecting-activating group for cysteine, particularly in the synthesis of cyclic and unsymmetrical disulfides using the Boc strategy. This chemistry has been extended to peptides assembled by the Fmoc strategy. N-Terminal
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.