추천 제품
분석
99%
형태
liquid
refractive index
n20/D 1.4611 (lit.)
bp
198-200 °C (lit.)
density
1.235 g/mL at 25 °C (lit.)
작용기
fluoro
ketone
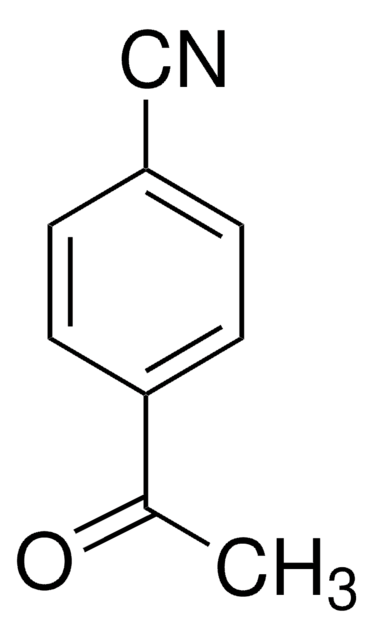
SMILES string
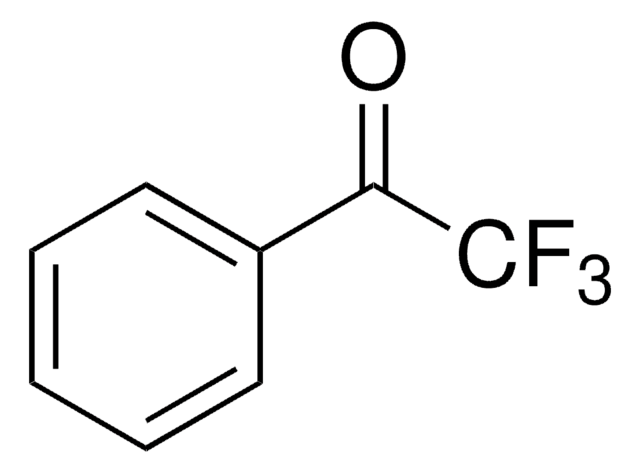
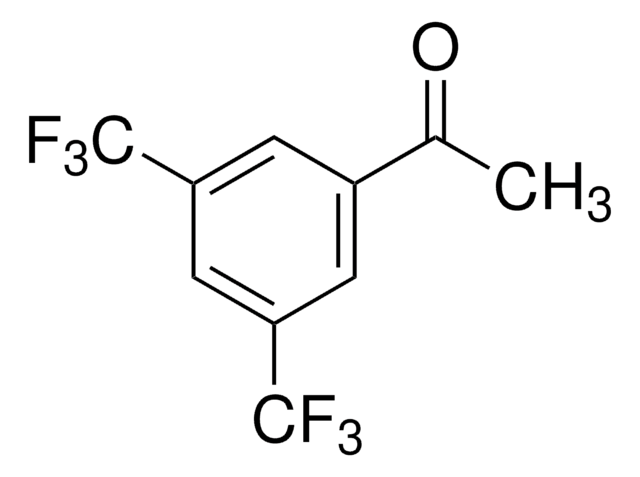
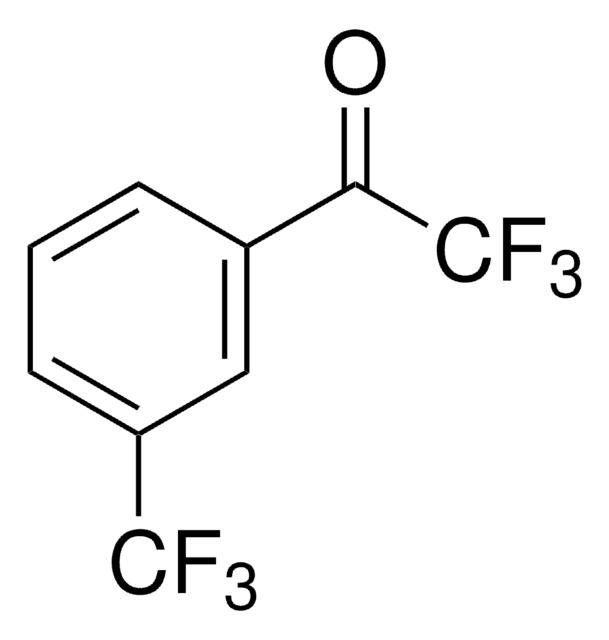
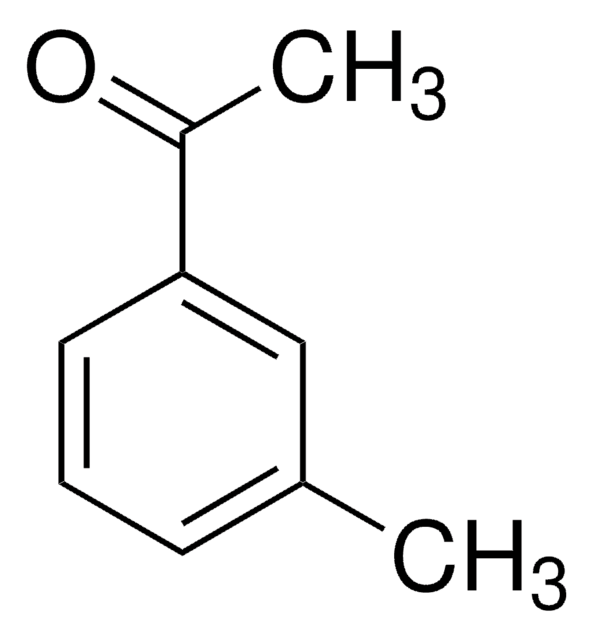
CC(=O)c1cccc(c1)C(F)(F)F
InChI
1S/C9H7F3O/c1-6(13)7-3-2-4-8(5-7)9(10,11)12/h2-5H,1H3
InChI key
ABXGMGUHGLQMAW-UHFFFAOYSA-N
유사한 제품을 찾으십니까? 방문 제품 비교 안내
일반 설명
Asymmetric catalytic addition of ethyl groups to 3′-(trifluoromethyl)acetophenone catalyzed by ligands derived from trans-1,2-diaminocyclohexane and camphor sulfonyl chloride has been reported. Phenylation of 3′-(trifluoromethyl)acetophenone in the presence of dihydroxy bis(sulfonamide) ligand (enantioselective catalyst), titanium tetraisopropoxide and diphenylzinc has been investigated.
애플리케이션
3′-(Trifluoromethyl)acetophenone has been used in a key step during the preparation of a commercial fungicide.
신호어
Warning
유해 및 위험 성명서
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
표적 기관
Respiratory system
Storage Class Code
10 - Combustible liquids
WGK
WGK 1
Flash Point (°F)
174.2 °F - closed cup
Flash Point (°C)
79 °C - closed cup
개인 보호 장비
Eyeshields, Gloves, type ABEK (EN14387) respirator filter
이미 열람한 고객
Serafino Gladiali et al.
Chemical Society reviews, 35(3), 226-236 (2006-03-01)
Hydrogen transfer reduction processes are attracting increasing interest from synthetic chemists in view of their operational simplicity and high selectivity. In this tutorial review the most significant advances recently achieved in the stereoselective reduction of unsaturated organic compounds catalyzed by
Celina García et al.
Journal of the American Chemical Society, 124(37), 10970-10971 (2002-09-13)
Many catalysts will promote the asymmetric addition of alkylzinc reagents to aldehydes. In contrast, there are no reports of additions to ketones that are both general and highly enantioselective. We describe herein a practical catalytic asymmetric addition of ethyl groups
Celina García et al.
Organic letters, 5(20), 3641-3644 (2003-09-26)
[reaction: see text] The catalytic asymmetric addition of phenyl groups from diphenylzinc to ketones is reported. The catalyst, generated from a dihydroxy bis(sulfonamide) ligand and titanium tetraisopropoxide, gives good to excellent enantioselectivities with a range of substrates.
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.