모든 사진(3)
About This Item
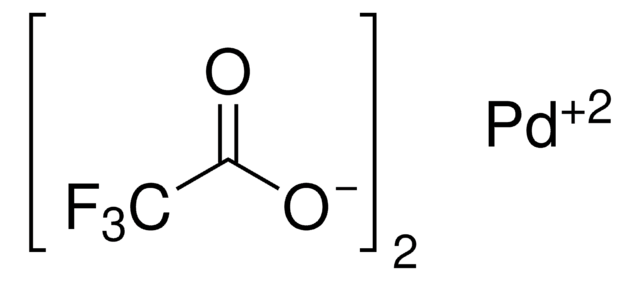
Linear Formula:
Pd(OCOCH3)2
CAS Number:
Molecular Weight:
224.51
Beilstein:
6086766
EC Number:
MDL number:
UNSPSC 코드:
12161600
PubChem Substance ID:
NACRES:
NA.22
추천 제품
Grade
reagent grade
Quality Level
분석
98%
양식
powder
반응 적합성
core: palladium
reaction type: Buchwald-Hartwig Cross Coupling Reaction
reaction type: Heck Reaction
reaction type: Hiyama Coupling
reaction type: Negishi Coupling
reaction type: Sonogashira Coupling
reaction type: Stille Coupling
reaction type: Suzuki-Miyaura Coupling
reagent type: catalyst
mp
216.3-223.7 °C (dec.)
SMILES string
CC(O[Pd]OC(C)=O)=O
InChI
1S/2C2H4O2.Pd/c2*1-2(3)4;/h2*1H3,(H,3,4);/q;;+2/p-2
InChI key
YJVFFLUZDVXJQI-UHFFFAOYSA-L
유사한 제품을 찾으십니까? 방문 제품 비교 안내
일반 설명
[Pd(OAc)2]3 is a palladium coordination complex and a heterogenous metal catalyst that is widely used in organic synthesis such as transmetalation, insertion, oxidative addition, direct homocoupling of aryl halides, Buchwald-Hartwig reaction of C-N bond formation, reduction of alkynes, and reductive elimination reactions. It is also employed as a starting material for the synthesis of other Pd(II) compounds as well as for the preparation of active palladium catalysts for Suzuki-Miyaura cross-coupling and C-H functionalization reactions.
애플리케이션
[Pd(OAc)2]3 can be used as a:
- Catalyst for the regioselective anti-hydrochlorination of the terminal and internal alkynes.
- Precursor to prepare a heterogeneous palladium complex catalyst for the Heck-Coupling reaction and Sonogashira cross-coupling reaction.
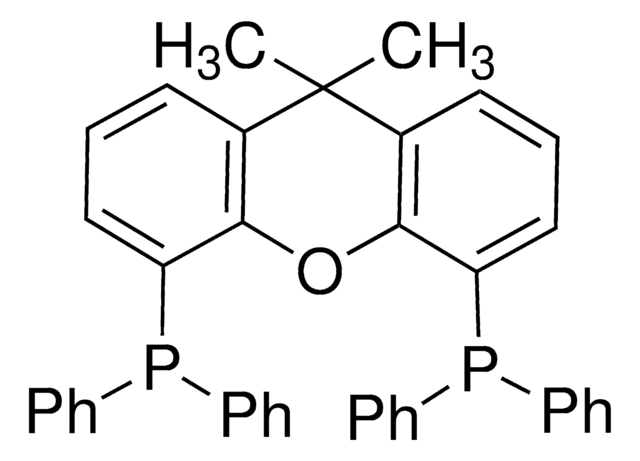
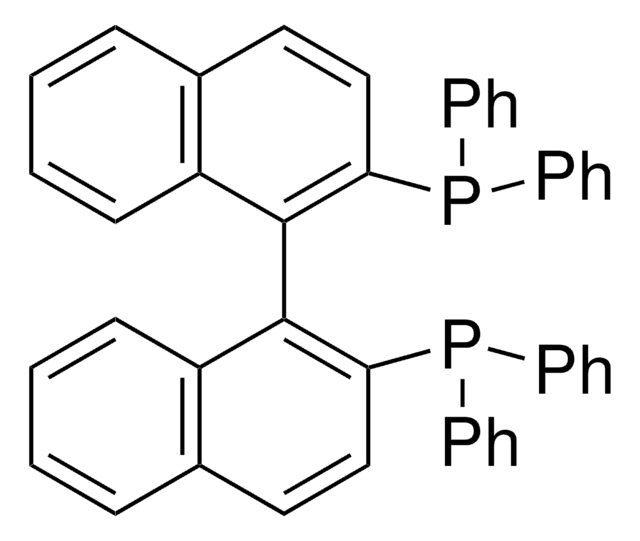
관련 제품
신호어
Danger
유해 및 위험 성명서
Hazard Classifications
Aquatic Acute 1 - Aquatic Chronic 1 - Eye Dam. 1 - Skin Sens. 1A
Storage Class Code
11 - Combustible Solids
WGK
WGK 2
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
개인 보호 장비
dust mask type N95 (US), Eyeshields, Gloves
이미 열람한 고객
Mark McLaughlin et al.
Organic letters, 8(15), 3311-3314 (2006-07-14)
[Structure: see text] An efficient regioselective method for the preparation of structurally diverse imidazopyridinones and benzoimidazolones starting from readily available and economical starting materials is described. High-yielding reductive alkylation of electron-deficient o-haloarylamines followed by treatment with inexpensive N-chlorosulfonyl isocyanate afforded
Wolfe, J. P.; Buchwald, S. L.
The Journal of Organic Chemistry, 62, 1264-1264 (1997)
Mark S Chen et al.
Journal of the American Chemical Society, 126(5), 1346-1347 (2004-02-05)
Sulfoxide ligation to Pd(II) salts is shown to selectively promote C-H oxidation versus Wacker oxidation chemistry and to control the regioselectivity in the C-H oxidation products. A catalytic method for the direct C-H oxidation of monosubstituted olefins to linear (E)-allylic
Ikuyo Kamiya et al.
The Journal of organic chemistry, 70(2), 696-698 (2005-01-18)
A highly regioselective hydroselenation of terminal alkynes with benzeneselenol can be achieved by the combination of palladium acetate and pyridine, providing the corresponding terminal alkenes, (i.e., 2-phenylseleno-1-alkenes) as a sole product. In this hydroselenation, pyridine may act as a suitable
Brunner, M.; Alper, H
The Journal of Organic Chemistry, 62, 7565-7565 (1997)
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.



![[1,1′-Bis(diphenylphosphino)ferrocene]dichloropalladium(II)](/deepweb/assets/sigmaaldrich/product/structures/130/734/8846aa26-1858-458a-998d-8c306c13bf0f/640/8846aa26-1858-458a-998d-8c306c13bf0f.png)