모든 사진(3)
About This Item
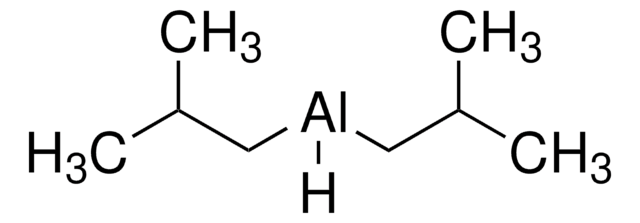
Linear Formula:
[(CH3)2CHCH2]2AlH
CAS Number:
Molecular Weight:
142.22
Beilstein:
4123663
MDL number:
UNSPSC 코드:
12352001
PubChem Substance ID:
NACRES:
NA.22
추천 제품
일반 설명
Diisobutylaluminum hydride reducing agent is commonly used for the reduction of an ester to aldehyde.
애플리케이션
Diisobutylaluminum hydride solution (1.0 M in hexanes) can be used in the synthesis of potassium diisobutyl-t-butoxyaluminum hydride (PDBBA), a novel reducing agent for the chemoselective conversion of esters in the presence of nitriles. The DIBAL-triethylamine complex can reduce α-oxoketene dithioacetal to the saturated ketone.
Used in Pd-catalyzed reductive debromination of secondary alkyl bromides. O-Debenzylation and ring opening of perbenzylated furanosides. Convenient in situ generation of HZrCp2Cl from ZrCp2Cl2 and DIBAL-H.
신호어
Danger
Hazard Classifications
Aquatic Chronic 2 - Asp. Tox. 1 - Eye Dam. 1 - Flam. Liq. 2 - Pyr. Liq. 1 - Repr. 2 - Skin Corr. 1B - STOT RE 1 Inhalation - STOT SE 3 - Water-react 1
표적 기관
Central nervous system, Nervous system
보충제 위험성
Storage Class Code
4.2 - Pyrophoric and self-heating hazardous materials
WGK
WGK 3
Flash Point (°F)
-9.4 °F - closed cup
Flash Point (°C)
-23 °C - closed cup
이미 열람한 고객
alpha.-Oxoketene dithioacetal chemistry. 2. Conjugate reductions with electrophilic reducing agents.
Gammill R
The Journal of Organic Chemistry, 46(17), 3555-3558 (1981)
Chemoselective Reduction of Esters to Aldehydes by Potassium Diisobutyl-t-butoxyaluminum Hydride (PDBBA).
Chae MJ, et al.
Bull. Korean Chem. Soc., 28(12), 2517-2517 (2007)
Damien Webb et al.
Organic letters, 14(2), 568-571 (2011-12-31)
A continuous flow system for the multiparameter (flow rate, temperature, residence time, stoichiometry) optimization of the DIBALH reduction of esters to aldehydes is described. Incorporating an in-line quench (MeOH), these transformations are generally complete in fewer than 60 s. Mixing
Hidetsura Cho et al.
The Journal of organic chemistry, 75(3), 627-636 (2009-12-31)
A systematic investigation of the reductive ring-expansion reaction of cyclic ketoximes fused to aromatic rings with diisobutylaluminum hydride (DIBALH) is described. This reaction regioselectively afforded a variety of five- to eight-membered bicyclic heterocycles or tricyclic heterocycles containing nitrogen neighboring an
D J Kopecky et al.
The Journal of organic chemistry, 65(1), 191-198 (2000-05-18)
An optimized protocol for the DIBALH reductive acetylation of acyclic esters and diesters is described. This reductive acetylation procedure allows a wide variety of esters to be converted into the corresponding alpha-acetoxy ethers in good to excellent yields. It was
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.