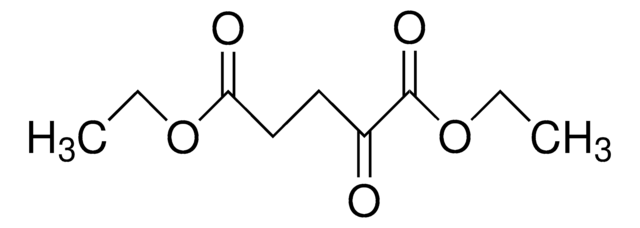
171263
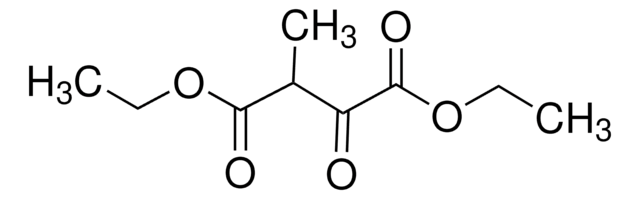
Diethyl oxalacetate sodium salt
95%
동의어(들):
Diethyl oxalacetate, Diethyl oxaloacetate, Diethyl oxosuccinate sodium salt, Oxalacetic acid diethyl ester sodium salt, Sodium diethyl oxalacetate
로그인조직 및 계약 가격 보기
모든 사진(3)
About This Item
Linear Formula:
C2H5OCOC(ONa)=CHCOOC2H5
CAS Number:
Molecular Weight:
210.16
Beilstein:
4279609
EC Number:
MDL number:
UNSPSC 코드:
12352100
PubChem Substance ID:
NACRES:
NA.22
추천 제품
Quality Level
분석
95%
mp
188-190 °C (lit.)
작용기
ester
SMILES string
[Na+].CCOC(=O)\C=C(/[O-])C(=O)OCC
InChI
1S/C8H12O5.Na/c1-3-12-7(10)5-6(9)8(11)13-4-2;/h5,9H,3-4H2,1-2H3;/q;+1/p-1/b6-5-;
InChI key
UJZUICGIJODKOS-YSMBQZINSA-M
애플리케이션
Diethyl oxalacetate sodium salt can be used as a reagent to synthesize:
- Spiro[indoline-3,4′-pyrano[2,3-c]pyrazole derivatives via a one-pot reaction with isatins, malononitrile, and hydrazine hydrate.
- 2,3-dioxo-4-carboxy-5-substituted pyrrolidines by reacting with various aldehydes and primary amines.
- 2-nitro-4-fluoro triazole diester by treating with 2-nitro-4-fluoro-phenylazide.
신호어
Warning
유해 및 위험 성명서
Hazard Classifications
Acute Tox. 4 Oral - Eye Irrit. 2 - Skin Irrit. 2
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
개인 보호 장비
dust mask type N95 (US), Eyeshields, Gloves
이미 열람한 고객
New 1, 2, 3-triazolo [1, 5-a] quinoxalines: synthesis and binding to benzodiazepine and adenosine receptors. II
Biagi G, et al.
European Journal of Medicinal Chemistry, 37(7), 565-571 (2002)
PYRUVATE CARBOXYLASE. I. NATURE OF THE REACTION.
M F UTTER et al.
The Journal of biological chemistry, 238, 2603-2608 (1963-08-01)
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.