모든 사진(1)
About This Item
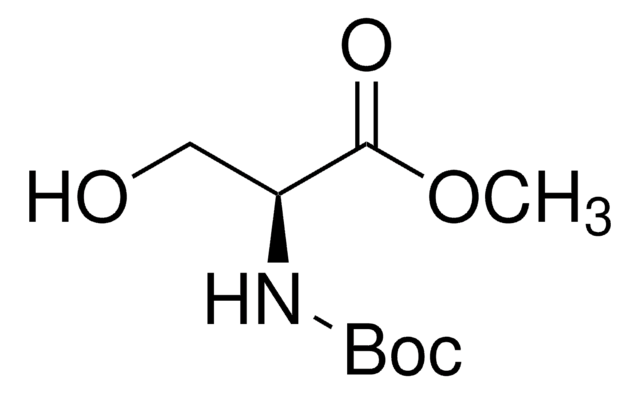
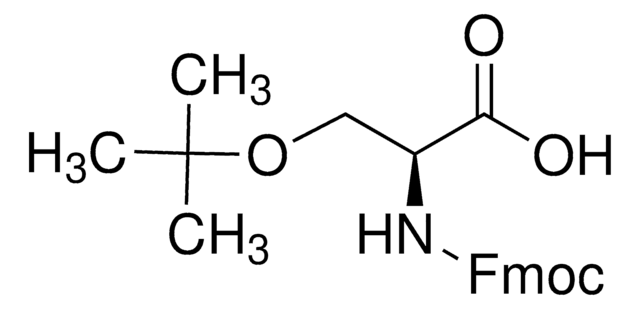
Linear Formula:
HOCH2CH(COOH)NHCOOC(CH3)3
CAS Number:
Molecular Weight:
205.21
Beilstein:
2212252
EC Number:
MDL number:
UNSPSC 코드:
12352209
eCl@ss:
32160406
PubChem Substance ID:
NACRES:
NA.22
추천 제품
Quality Level
분석
≥99.0% (T)
광학 활성
[α]20/D −3.5±0.5°, c = 2% in acetic acid
반응 적합성
reaction type: Boc solid-phase peptide synthesis
mp
91 °C (dec.) (lit.)
응용 분야
peptide synthesis
저장 온도
2-8°C
SMILES string
CC(C)(C)OC(=O)N[C@@H](CO)C(O)=O
InChI
1S/C8H15NO5/c1-8(2,3)14-7(13)9-5(4-10)6(11)12/h5,10H,4H2,1-3H3,(H,9,13)(H,11,12)/t5-/m0/s1
InChI key
FHOAKXBXYSJBGX-YFKPBYRVSA-N
유사한 제품을 찾으십니까? 방문 제품 비교 안내
일반 설명
Boc-Ser-OH is an amino acid derivative.
애플리케이션
Boc-Ser-OH may be used in the synthesis of the following:
- 2-(N-Fmoc)-3-(N-Boc-N-methoxy)-diaminopropanoic acid (Fmoc: 9-fluorenylmethoxycarbonyl; Boc: t-butyloxycarbonyl)
- Boc-Ser-Leu-OMe
- cyclic peptide synthesis
- benzylsulfonyl-D-Ser-Ser-4-amidinobenzylamide
Building block in peptide synthesis. Starting material for the synthesis of various α-amino acids via the β-lactone.
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
개인 보호 장비
Eyeshields, Gloves, type N95 (US)
이미 열람한 고객
Synthesis of cyclic peptides via O-N-acyl migration.
Lecaillon J, et al.
Tetrahedron Letters, 49(31), 4674-4676 (2008)
The relationship between taste and primary structure of ?delicious peptide?(Lys-Gly-Asp-Glu-Glu-Ser-Leu-Ala) from beef soup.
Tamura M, et al.
Agricultural and Biological Chemistry, 53(2), 319-325 (1989)
Michael R Carrasco et al.
Biopolymers, 84(4), 414-420 (2006-03-02)
Amino acids with N-alkylaminooxy side chains have proven effective for the rapid synthesis of neoglycopeptides. Chemoselective reaction of reducing sugars with peptides containing these amino acids provides glycoconjugates that are structurally similar to their natural counterparts. 2-(N-Fmoc)-3-(N-Boc-N-methoxy)-diaminopropanoic acid (Fmoc: 9-fluorenylmethoxycarbonyl;
O García-Suárez et al.
Immunology, 94(2), 235-241 (1998-09-19)
Increasing evidence suggests that some members of the neurotrophic factor family of neurotrophins could be implicated in the regulation of immune responses. Neurotrophins, as well as their tyrosine kinase signal-transducing receptors (the so-called Trk neurotrophin receptors), have been detected in
Gema Villa-Fombuena et al.
Development (Cambridge, England), 148(18) (2021-08-10)
Drosophila female germline stem cells (GSCs) are found inside the cellular niche at the tip of the ovary. They undergo asymmetric divisions to renew the stem cell lineage and to produce sibling cystoblasts that will in turn enter differentiation. GSCs
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.