138797
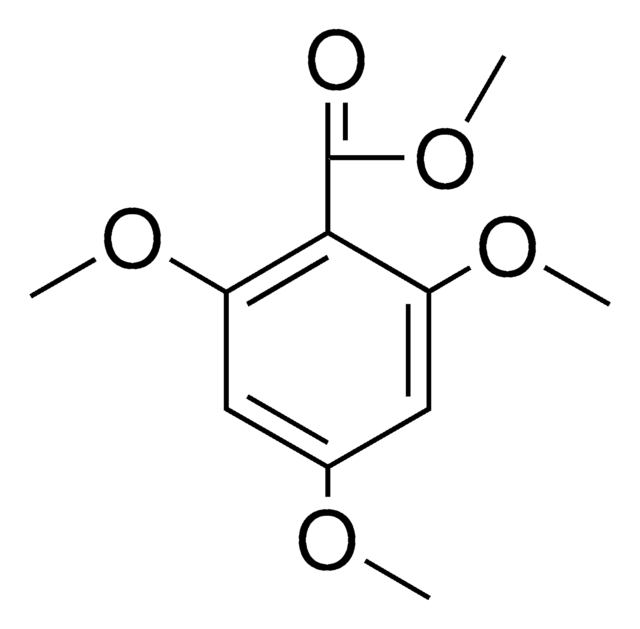
4,6-Dimethoxysalicylaldehyde
98%
동의어(들):
2,4-Dimethoxy-6-hydroxybenzaldehyde, 4,6-Dimethoxy-2-hydroxybenzaldehyde
로그인조직 및 계약 가격 보기
모든 사진(1)
About This Item
Linear Formula:
HOC6H2(OCH3)2CHO
CAS Number:
Molecular Weight:
182.17
Beilstein:
1241679
EC Number:
MDL number:
UNSPSC 코드:
12352100
PubChem Substance ID:
NACRES:
NA.22
추천 제품
분석
98%
mp
68-70 °C (lit.)
작용기
aldehyde
SMILES string
[H]C(=O)c1c(O)cc(OC)cc1OC
InChI
1S/C9H10O4/c1-12-6-3-8(11)7(5-10)9(4-6)13-2/h3-5,11H,1-2H3
InChI key
FQRQWPNYJOFDLO-UHFFFAOYSA-N
유사한 제품을 찾으십니까? 방문 제품 비교 안내
일반 설명
4,6-Dimethoxysalicylaldehyde on condensation with methylamine yields Schiff bases.
애플리케이션
4,6-Dimethoxysalicylaldehyde was used in the preparation of a new class of efficient ketocoumarin triplet sensitizers. It was used as staring reagent in the total synthesis of (+/-)-linderol A, a hexahydrodibenzofuran.
생화학적/생리학적 작용
4,6-Dimethoxysalicylaldehyde has antimicrobial activity against Candida albicans.
신호어
Warning
유해 및 위험 성명서
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
표적 기관
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
개인 보호 장비
dust mask type N95 (US), Eyeshields, Gloves
이미 열람한 고객
Wojciech Schilf et al.
Magnetic resonance in chemistry : MRC, 42(6), 556-560 (2004-05-12)
Hydrogen bonding within the structures of three Schiff bases (1-3), obtained by condensation of 4-methoxy-, 5-methoxy- and 4,6-dimethoxysalicylaldehyde with methylamine, was investigated by measuring deuterium and tritium NMR isotope effects. The primary deuterium and tritium isotope effects (delta(XH)-delta(XD/T)) and secondary
Eila Pelttari et al.
Zeitschrift fur Naturforschung. C, Journal of biosciences, 62(7-8), 487-497 (2007-10-05)
A systematic survey of the antimicrobial properties of substituted salicylaldehydes and some related aromatic aldehydes is reported. A total of 23 different compounds, each at four different concentrations, were studied using a panel of seven microbes (Aspergillus niger, Bacillus cereus
Ketocoumarins: a new class of triplet sensitizers.
Specht DP, et al.
Tetrahedron, 38(9), 1203-1211 (1982)
M Yamashita et al.
Organic letters, 3(9), 1359-1362 (2001-05-12)
[reaction in text] The first total synthesis of (+/-)-linderol A, a hexahydrodibenzofuran isolated from Lindera umbellata bark, with potent inhibitory activity on melanin biosynthesis of cultured B-16 melanoma cells was achieved via a 20-step of reaction in 7.64% overall yield
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.![2,3,6,7-Tetrahydro-8-hydroxy-1H,5H-benzo[ij]quinolizine-9-carboxaldehyde 98%](/deepweb/assets/sigmaaldrich/product/structures/166/830/a0d9a84a-5623-41a1-a54b-3b0272e5b28c/640/a0d9a84a-5623-41a1-a54b-3b0272e5b28c.png)